Przejdź do
Zaloguj się, aby wyświetlić ceny organizacyjne i kontraktowe.
Wybierz wielkość
Zmień widok
| Gabaryty przesyłki | SKU | Dostępność | Cena netto |
|---|---|---|---|
| 25 mg | Skontaktuj się z Obsługą Klienta, aby uzyskać informacje na temat dostępności | 1000,00 zł |
Informacje o tej pozycji
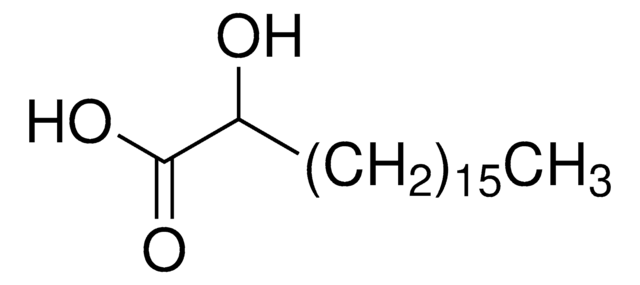
Wzór empiryczny (zapis Hilla):
C18H36O3
Numer CAS:
Masa cząsteczkowa:
300.48
NACRES:
NA.25
PubChem Substance ID:
UNSPSC Code:
12352211
MDL number:
1000,00 zł
Skontaktuj się z Obsługą Klienta, aby uzyskać informacje na temat dostępności
Pomoc techniczna
Potrzebujesz pomocy? Nasz zespół doświadczonych naukowców chętnie Ci pomoże.
Pozwól nam pomócbiological source
synthetic (organic)
Quality Level
assay
≥99%
form
powder
functional group
carboxylic acid
lipid type
saturated FAs
shipped in
ambient
storage temp.
2-8°C
SMILES string
CCCCCCCCCCCCCCCCC(O)C(O)=O
InChI
1S/C18H36O3/c1-2-3-4-5-6-7-8-9-10-11-12-13-14-15-16-17(19)18(20)21/h17,19H,2-16H2,1H3,(H,20,21)
InChI key
KIHBGTRZFAVZRV-UHFFFAOYSA-N
Biochem/physiol Actions
DL-α-Hydroxystearic acid is a mixture of D and L-α-hydroxystearic acid (2-Hydroxyoctadecanoic acid) enantiomers. α-Hydroxystearic may be used in studies on the properties and metabolism of α-hydroxylated (2-hydroxylated) medium chain fatty acids.
Packaging
Sealed ampule.
Ta strona może zawierać tekst przetłumaczony maszynowo.
1 of 1
Ta pozycja | |||
|---|---|---|---|
| functional group carboxylic acid | functional group carboxylic acid | functional group carboxylic acid | functional group ester |
| assay ≥99% | assay ≥98% | assay ≥99% (GC) | assay ≥99% (GC) |
| biological source synthetic (organic) | biological source synthetic (organic) | biological source synthetic (organic) | biological source - |
| storage temp. 2-8°C | storage temp. 2-8°C | storage temp. 2-8°C | storage temp. −20°C |
| form powder | form powder | form powder | form powder |
| shipped in ambient | shipped in ambient | shipped in ambient | shipped in ambient |
Klasa składowania
11 - Combustible Solids
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
Eyeshields, Gloves, type N95 (US)
Wybierz jedną z najnowszych wersji:
Masz już ten produkt?
Dokumenty związane z niedawno zakupionymi produktami zostały zamieszczone w Bibliotece dokumentów.
Powiązane treści
M Salim-Hanna et al.
Lipids, 24(8), 750-752 (1989-08-01)
Combined--but not individual--microsomal and supernatant fractions obtained from rat brains not only consume oxygen but also provoke emission from added chlorophyll. These results are consistent with literature data (Levis and Mead, J. Biol. Chem. 239, 77 [1964]) for trapping of
Conrad D Lendrum et al.
Langmuir : the ACS journal of surfaces and colloids, 27(8), 4430-4438 (2011-03-23)
2-Hydroxyacids display complex monolayer phase behavior due to the additional hydrogen bonding afforded by the presence of the second hydroxy group. The placement of this group at the position α to the carboxylic acid functionality also introduces the possibility of
R Schütz et al.
International journal of cosmetic science, 41(3), 240-256 (2019-04-08)
We report on the preparation and efficacy of 10-hydroxystearic acid (HSA) that improves facial age spots and conspicuous pores. The hydration of oleic acid into HSA was catalyzed by the oleate hydratase from Escherichia coli. Following treatment with HSA, collagen
Numer pozycji handlu globalnego
| SKU | NUMER GTIN |
|---|---|
| H9631-25MG | 04061833812785 |