Wybierz wielkość
| Gabaryty przesyłki | SKU | Dostępność | Cena netto |
|---|---|---|---|
| 10 mg | Sprawdź dostępność w koszyku | 495,00 zł |
Informacje o tej pozycji
495,00 zł
Quality Level
description
Merck USA index - 14, 3548
assay
≥99% (HPLC)
form
solid
manufacturer/tradename
Calbiochem®
storage condition
OK to freeze
color
bright yellow
solubility
ethanol: 1 mg/mL, DMSO: 5 mg/mL
shipped in
ambient
storage temp.
2-8°C
SMILES string
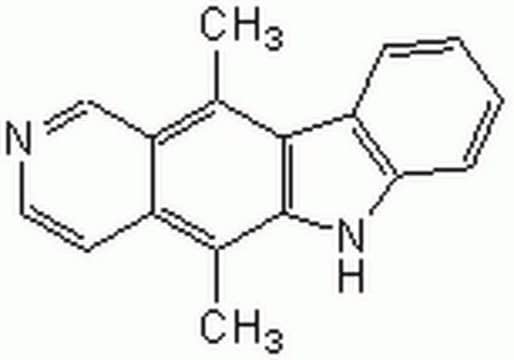
[nH]1c2c(c4c1cccc4)c(c3c(c2C)ccnc3)C
InChI
1S/C17H14N2/c1-10-14-9-18-8-7-12(14)11(2)17-16(10)13-5-3-4-6-15(13)19-17/h3-9,19H,1-2H3
InChI key
CTSPAMFJBXKSOY-UHFFFAOYSA-N
General description
Biochem/physiol Actions
topoisomerase 2
Preparation Note
Other Notes
Schwaller, M.A., et al. 1995. J. Biol. Chem.270, 22709.
Pommier, Y., et al. 1985. Biochemistry24, 6406.
Legal Information
Disclaimer
1 of 1
Ta pozycja | |||
|---|---|---|---|
| assay ≥99% (HPLC) | assay ≥95% (HPLC) | assay ≥98% (TLC) | assay ≥95% (HPLC) |
| form solid | form solid | form solid | form solid |
| Quality Level 100 | Quality Level 100 | Quality Level 100 | Quality Level 100 |
| manufacturer/tradename Calbiochem® | manufacturer/tradename Calbiochem® | manufacturer/tradename Calbiochem® | manufacturer/tradename Calbiochem® |
| storage temp. 2-8°C | storage temp. 2-8°C | storage temp. 2-8°C | storage temp. 2-8°C |
| storage condition OK to freeze | storage condition OK to freeze | storage condition OK to freeze, protect from light | storage condition OK to freeze, protect from light |
signalword
Danger
hcodes
Hazard Classifications
Acute Tox. 3 Oral
Klasa składowania
6.1C - Combustible acute toxic Cat.3 / toxic compounds or compounds which causing chronic effects
wgk
WGK 3
Certyfikaty analizy (CoA)
Poszukaj Certyfikaty analizy (CoA), wpisując numer partii/serii produktów. Numery serii i partii można znaleźć na etykiecie produktu po słowach „seria” lub „partia”.
Masz już ten produkt?
Dokumenty związane z niedawno zakupionymi produktami zostały zamieszczone w Bibliotece dokumentów.
Powiązane treści
Numer pozycji handlu globalnego
| SKU | NUMER GTIN |
|---|---|
| 324688-10MG | 04055977196863 |