Wybierz wielkość
| Gabaryty przesyłki | SKU | Dostępność | Cena netto |
|---|---|---|---|
| 100 μg | Przewidywany termin wysyłki28 maja 2026zKuehne + Nagel Sp. z o.o. | 2240,00 zł |
Informacje o tej pozycji
Quality Segment
biological source
rabbit
antibody form
purified antibody
antibody product type
primary antibodies
clone
RM146, monoclonal
species reactivity (predicted by homology)
all
technique(s)
immunoprecipitation (IP): suitable, western blot: suitable
isotype
IgG
shipped in
wet ice
target post-translational modification
unmodified
Gene Information
human ... HRG(3273)
General description
Immunogen
Application
Przeciwciała drugorzędowe i kontrolne
Przeciwciała drugorzędowe adsorbowane w celu podwójnego znakowania
Biochem/physiol Actions
Physical form
Preparation Note
Uwaga: Zmienność temperatur w zamrażarce poniżej -20°C może powodować zamarzanie roztworów zawierających glicerol podczas przechowywania.
Analysis Note
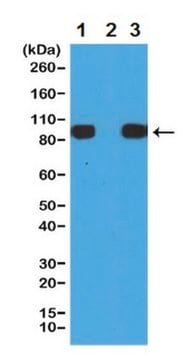
Western Blotting Analysis (WB): 0.2 μg/mL of this antibody detected Histidine Tag in HEK293T cell lysate transfected with His-Tag fusion protein X and E. coli lysate with His-Tag Protein Y.
Other Notes
Disclaimer
1 of 1
Ta pozycja | |||
|---|---|---|---|
| antibody form purified antibody | antibody form affinity purified immunoglobulin | antibody form purified antibody | antibody form purified antibody |
| biological source rabbit | biological source mouse | biological source mouse | biological source mouse |
| clone RM146, monoclonal | clone 4D11, monoclonal | clone DG122-2A7, monoclonal | clone HIS.H8, monoclonal |
| technique(s) immunoprecipitation (IP): suitable, western blot: suitable | technique(s) immunocytochemistry: suitable, western blot: suitable | technique(s) immunoprecipitation (IP): suitable, western blot: suitable | technique(s) immunocytochemistry: suitable, immunoprecipitation (IP): suitable, western blot: suitable |
| shipped in wet ice | shipped in wet ice | shipped in dry ice | shipped in dry ice |
| Gene Information human ... HRG(3273) | Gene Information human ... HRG(3273) | Gene Information - | Gene Information - |
Still not finding the right product?
Wypróbuj nasze narzędzie Narzędzie selektora produktów, aby zawęzić opcje.
Klasa składowania
12 - Non Combustible Liquids
wgk
WGK 2
flash_point_f
does not flash
flash_point_c
does not flash
Certyfikaty analizy (CoA)
Poszukaj Certyfikaty analizy (CoA), wpisując numer partii/serii produktów. Numery serii i partii można znaleźć na etykiecie produktu po słowach „seria” lub „partia”.
Masz już ten produkt?
Dokumenty związane z niedawno zakupionymi produktami zostały zamieszczone w Bibliotece dokumentów.
Powiązane treści
A major focus of breast cancer research is to understand the mechanisms responsible for disease progression and drug resistance. Toward that end, it has been found that approximately two thirds of all human breast carcinomas overexpress the Estrogen Receptor α (ERα) protein and it remains the primary pharmacological target for endocrine therapy1,2. The normal cellular function of ERα is as a transcription factor that mediates a wide variety of physiological processes, many of which are dependent upon phosphorylation of the receptor at specific amino acid residues3,4. Indeed, ERα is known to be phosphorylated at a multitude of different sites, yet how these all correlate to disease remains unclear5. Here, we interrogated multiple sites of ERα for phosphorylation status by screening an extensive panel of different breast cancer patient samples and other non-breast cancer tissue microarray (TMA) slide samples to determine their relevance to disease.