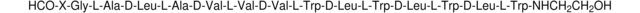A4665
Alamethicin from Trichoderma viride
≥98% (HPLC)
Synonim(y):
Antibiotic U-22324
About This Item
Polecane produkty
Poziom jakości
Próba
≥98% (HPLC)
Postać
powder
spektrum działania antybiotyku
Gram-positive bacteria
Tryb działania
cell membrane | interferes
temp. przechowywania
2-8°C
ciąg SMILES
CC(C)C[C@H](NC(=O)CNC(=O)C(C)(C)NC(=O)[C@@H](NC(=O)C(C)(C)NC(=O)[C@H](CCC(N)=O)NC(=O)[C@H](C)NC(=O)C(C)(C)NC(=O)[C@H](C)NC(=O)C(C)(C)NC(=O)[C@@H]1CCCN1C(=O)C(C)(C)NC(C)=O)C(C)C)C(=O)NC(C)(C)C(=O)N2CCC[C@H]2C(=O)N[C@@H](C(C)C)C(=O)NC(C)(C)C(=O)NC(C)(C)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](CO)Cc3ccccc3
InChI
1S/C92H150N22O25/c1-47(2)43-58(72(127)108-92(24,25)84(139)113-41-29-33-59(113)73(128)103-65(48(3)4)75(130)111-90(20,21)82(137)112-89(18,19)80(135)102-56(37-40-64(120)121)70(125)101-55(35-38-61(93)117)69(124)98-54(46-115)44-53-31-27-26-28-32-53)99-63(119)45-95-77(132)85(10,11)110-76(131)66(49(5)6)104-81(136)88(16,17)107-71(126)57(36-39-62(94)118)100-67(122)50(7)96-78(133)86(12,13)106-68(123)51(8)97-79(134)87(14,15)109-74(129)60-34-30-42-114(60)83(138)91(22,23)105-52(9)116/h26-28,31-32,47-51,54-60,65-66,115H,29-30,33-46H2,1-25H3,(H2,93,117)(H2,94,118)(H,95,132)(H,96,133)(H,97,134)(H,98,124)(H,99,119)(H,100,122)(H,101,125)(H,102,135)(H,103,128)(H,104,136)(H,105,116)(H,106,123)(H,107,126)(H,108,127)(H,109,129)(H,110,131)(H,111,130)(H,112,137)(H,120,121)/t50-,51-,54+,55-,56-,57-,58-,59-,60-,65-,66-/m0/s1
Klucz InChI
LGHSQOCGTJHDIL-SLKIUSOBSA-N
Szukasz podobnych produktów? Odwiedź Przewodnik dotyczący porównywania produktów
Powiązane kategorie
Opis ogólny
Zastosowanie
- to examine the effect of glutamate transporters on the Na+/K+-ATPase dependent extracellular K+ transient during neuronal activity
- to investigate the formation of anion-permeable channels in planar lipid bilayers by magainin I obtained from Xenopus skin
- to study the permeability of root apical meristem and epidermis and cellulase induced resistance to Alamethicin in Arabidopsis thaliana
- to study the cytotoxic effects of the phycotoxin okadaic acid and mycotoxins on human intestinal (HT-29) and neuroblastoma (SH-SY5Y) cell lines
- to analyze the structural variations and biological activity of peptaibols obtained from the Longibrachiatum clade belonging to the genus of filamentous fungi Trichoderma
Działania biochem./fizjol.
Jakość
Sekwencja
Hasło ostrzegawcze
Danger
Zwroty wskazujące rodzaj zagrożenia
Zwroty wskazujące środki ostrożności
Klasyfikacja zagrożeń
Acute Tox. 3 Oral
Kod klasy składowania
6.1C - Combustible acute toxic Cat.3 / toxic compounds or compounds which causing chronic effects
Klasa zagrożenia wodnego (WGK)
WGK 3
Temperatura zapłonu (°F)
Not applicable
Temperatura zapłonu (°C)
Not applicable
Środki ochrony indywidualnej
Eyeshields, Faceshields, Gloves, type P2 (EN 143) respirator cartridges
Certyfikaty analizy (CoA)
Poszukaj Certyfikaty analizy (CoA), wpisując numer partii/serii produktów. Numery serii i partii można znaleźć na etykiecie produktu po słowach „seria” lub „partia”.
Masz już ten produkt?
Dokumenty związane z niedawno zakupionymi produktami zostały zamieszczone w Bibliotece dokumentów.
Klienci oglądali również te produkty
Nasz zespół naukowców ma doświadczenie we wszystkich obszarach badań, w tym w naukach przyrodniczych, materiałoznawstwie, syntezie chemicznej, chromatografii, analityce i wielu innych dziedzinach.
Skontaktuj się z zespołem ds. pomocy technicznej