Wybierz wielkość
| Gabaryty przesyłki | SKU | Dostępność | Cena netto |
|---|---|---|---|
| 100 μg | Sprawdź dostępność w koszyku | 2110,00 zł |
Informacje o tej pozycji
biological source
mouse
Quality Level
antibody form
purified immunoglobulin
antibody product type
primary antibodies
clone
RL2, monoclonal
species reactivity (predicted by homology)
all
technique(s)
affinity binding assay: suitable, electron microscopy: suitable, immunocytochemistry: suitable, immunoprecipitation (IP): suitable, western blot: suitable
isotype
IgG1κ
shipped in
wet ice
target post-translational modification
unmodified
Gene Information
human ... OGT(8473)
General description
Immunogen
Application
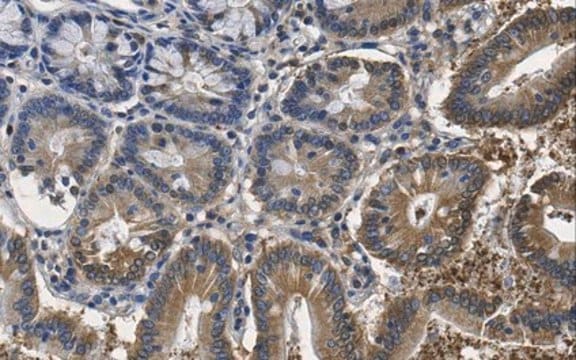
Immunocytochemistry Analysis: A representative lot immunostained nuclear envelopes, but not the nuclear interior, of digitonin-permeabilized HeLa cells. Clone RL2 stained the nuclear interior only among Triton X-100-permeabilized HeLa cells without intact nuclear envelopes (Adam, S.A., et al. (1990). J. Cell Biol. 111(3):807-816).
Affinity Binding Assay: A representative lot was radiolabeled with 125I and studied for its binding characteristics toward isolated rat liver nuclear envelopes (Snow, C.M., et al. (1987). J. Cell Biol. 104(5):1143-1156).
Electron Microscopy: A representative lot localized the O-GlcNAc immunoreactivity in isolated rat liver nuclear envelopes (Snow, C.M., et al. (1987). J. Cell Biol. 104(5):1143-1156).
Western Blotting Analysis: A representative lot detected O-GlcNAcylated proteins in rat liver nuclear envelopes preparations (Snow, C.M., et al. (1987). J. Cell Biol. 104(5):1143-1156; Holt, G.D., et al. (1987). J. Cell Biol. 104(5):1157-1164).
Immunoprecipitation Analysis: A representative lot immunoprecipitated O-GlcNAcylated proteins from solubilized rat liver nuclear envelopes preparations. Pretreatment of nuclear envelopes preparations with galactosyltrarnsferase prevented the immunoprecipitation of glycoproteins by clone RL2 (Snow, C.M., et al. (1987). J. Cell Biol. 104(5):1143-1156; Holt, G.D., et al. (1987). J. Cell Biol. 104(5):1157-1164).
Biochem/physiol Actions
Physical form
Analysis Note
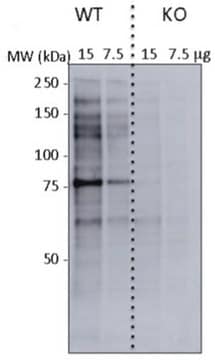
Western Blotting Analysis: 1.0 µg/mL of this antibody detected O-Linked N-Acetylglucosamine in 10 µg of HeLa cell lysate.
Other Notes
1 of 1
Ta pozycja | |||
|---|---|---|---|
| antibody form purified immunoglobulin | antibody form purified immunoglobulin | antibody form purified immunoglobulin | antibody form IgG fraction of antiserum |
| Gene Information human ... OGT(8473) | Gene Information human ... OGT(8473) | Gene Information human ... OGT(8473) | Gene Information human ... OGT(8473) |
| clone RL2, monoclonal | clone RL1, monoclonal | clone CTD110.6, monoclonal | clone polyclonal |
| biological source mouse | biological source mouse | biological source mouse | biological source rabbit |
| isotype IgG1κ | isotype IgMκ | isotype IgMκ | isotype - |
| shipped in wet ice | shipped in ambient | shipped in - | shipped in dry ice |
Still not finding the right product?
Wypróbuj nasze narzędzie Narzędzie selektora produktów, aby zawęzić opcje.
Klasa składowania
12 - Non Combustible Liquids
wgk
WGK 1
flash_point_f
Not applicable
flash_point_c
Not applicable
Certyfikaty analizy (CoA)
Poszukaj Certyfikaty analizy (CoA), wpisując numer partii/serii produktów. Numery serii i partii można znaleźć na etykiecie produktu po słowach „seria” lub „partia”.
Masz już ten produkt?
Dokumenty związane z niedawno zakupionymi produktami zostały zamieszczone w Bibliotece dokumentów.
Numer pozycji handlu globalnego
| SKU | NUMER GTIN |
|---|---|
| MABS157 | 04055977301243 |