Autenticati per visualizzare i prezzi organizzativi e contrattuali.
Scegli un formato
Cambia visualizzazione
| Taglio della confezione | SKU | Disponibilità | Prezzo |
|---|---|---|---|
| 10 g | Per informazioni sulla disponibilità, contatta il Servizio Clienti. | 119,00 € |
Informazioni su questo articolo
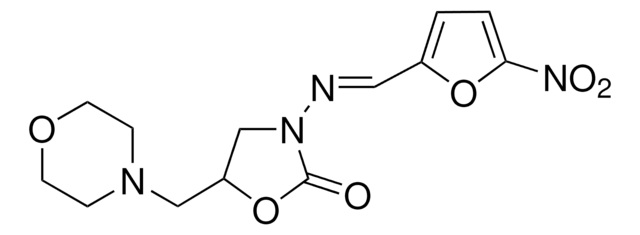
Formula empirica (notazione di Hill):
C8H7N3O5
Numero CAS:
Peso molecolare:
225.16
UNSPSC Code:
51102829
NACRES:
NA.85
PubChem Substance ID:
EC Number:
200-653-3
Beilstein/REAXYS Number:
8317414
MDL number:
119,00 €
Per informazioni sulla disponibilità, contatta il Servizio Clienti.
Servizio Tecnico
Hai bisogno di aiuto? Il nostro team di scienziati qualificati è a tua disposizione.
Permettici di aiutartibiological source
synthetic
Quality Segment
form
powder
color
yellow
mp
256-256 °C
solubility
formic acid: 50 mg/mL
antibiotic activity spectrum
Gram-positive bacteria, parasites
mode of action
enzyme | inhibits
SMILES string
[O-][N+](=O)c1ccc(\C=N\N2CCOC2=O)o1
InChI
1S/C8H7N3O5/c12-8-10(3-4-15-8)9-5-6-1-2-7(16-6)11(13)14/h1-2,5H,3-4H2/b9-5+
InChI key
PLHJDBGFXBMTGZ-WEVVVXLNSA-N
Application
Furazolidone is a nitrofuran derivative with antiprotozoal and antibacterial activity. Furazolidone binds bacterial DNA, which leads to the gradual inhibition of monoamine oxidase. It is used to treat anorexia and antagonism of thiamin utilization in poultry[1]. Furazolidone increases thapsigargin-sensitive Ca2+-ATPase in chick cardiac myocytes.
Biochem/physiol Actions
Furazolidone and its generated free radicals, may bind to DNA and induce cross-links. Bacterial DNA is particularly susceptible to this drug, which results in high levels of mutations (transitions and transversions) in the bacterial chromosome. Its mechanism of action minimizes the development of resistant organisms. Furazolidone is a monoamine oxidase (MAO) inhibitor and is used as a DNA interstrand cross-linking agent.
1 of 1
Questo articolo | |||
|---|---|---|---|
| mode of action enzyme | inhibits | mode of action - | mode of action - | mode of action - |
| antibiotic activity spectrum Gram-positive bacteria, parasites | antibiotic activity spectrum - | antibiotic activity spectrum - | antibiotic activity spectrum - |
| form powder | form - | form - | form solid |
| solubility formic acid: 50 mg/mL | solubility - | solubility - | solubility - |
| biological source synthetic | biological source - | biological source - | biological source - |
| color yellow | color - | color - | color - |
Still not finding the right product?
Explore all of our products under Furazolidone
signalword
Warning
hcodes
Hazard Classifications
Repr. 2
Classe di stoccaggio
11 - Combustible Solids
wgk
WGK 3
ppe
Eyeshields, Gloves, type P3 (EN 143) respirator cartridges
Scegli una delle versioni più recenti:
Possiedi già questo prodotto?
I documenti relativi ai prodotti acquistati recentemente sono disponibili nell’Archivio dei documenti.
Numero articolo commerciale globale
| SKU | GTIN |
|---|---|
| F9505-10G | 04061833621721 |
| F9505-25G | 04061833621776 |
| F9505-100G | 04061832991153 |