Fortfahren mit
Größe auswählen
| Packungsgröße | SKU | Verfügbarkeit | Preis |
|---|---|---|---|
| 10 mg | Warenkorb auf Verfügbarkeit prüfen | € 215,00 | |
| 25 mg | Warenkorb auf Verfügbarkeit prüfen | € 428,00 | |
| 100 mg | Warenkorb auf Verfügbarkeit prüfen | € 1.460,00 € 1.241,00 |
Über diesen Artikel
Produktname
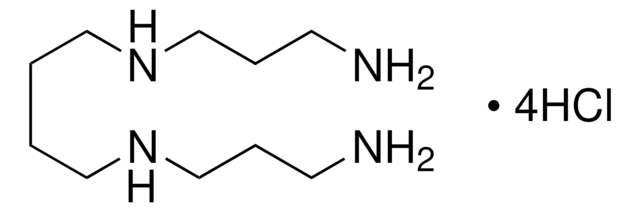
Methoctramine hydrate, ≥97% (NMR), solid
Quality Level
assay
≥97% (NMR)
form
solid
color
white
solubility
H2O: >20 mg/mL
storage temp.
2-8°C
SMILES string
O.Cl.Cl.Cl.Cl.COc1ccccc1CNCCCCCCNCCCCCCCCNCCCCCCNCc2ccccc2OC.COc3ccccc3CNCCCCCCNCCCCCCCCNCCCCCCNCc4ccccc4OC
InChI
1S/2C36H62N4O2.4ClH.H2O/c2*1-41-35-23-13-11-21-33(35)31-39-29-19-9-7-17-27-37-25-15-5-3-4-6-16-26-38-28-18-8-10-20-30-40-32-34-22-12-14-24-36(34)42-2;;;;;/h2*11-14,21-24,37-40H,3-10,15-20,25-32H2,1-2H3;4*1H;1H2
InChI key
XIIINYPADNNZHA-UHFFFAOYSA-N
Gene Information
human ... CHRM2(1129)
General description
Application
- As a muscarinic receptor 2 antogonist.
- As a test molecule to check its effect on premature osteogenic differentiation of HGPS (Hutchinson-Gilford progeria syndrome) iPS (induced pluripotent)-derived mesenchymal stem cells.
Biochem/physiol Actions
1 of 1
Dieser Artikel | |||
|---|---|---|---|
| assay ≥97% (NMR) | assay ≥99.0% (AT) | assay - | assay - |
| Quality Level 100 | Quality Level 300 | Quality Level 300 | Quality Level 200 |
| form solid | form powder or crystals | form solid | form powder or crystals |
| Gene Information human ... CHRM2(1129) | Gene Information - | Gene Information - | Gene Information human ... GRIN1(2902) |
| storage temp. 2-8°C | storage temp. room temp | storage temp. room temp | storage temp. room temp |
| color white | color - | color - | color - |
Lagerklasse
11 - Combustible Solids
wgk
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
Eyeshields, Gloves, type N95 (US)
Hier finden Sie alle aktuellen Versionen:
Besitzen Sie dieses Produkt bereits?
In der Dokumentenbibliothek finden Sie die Dokumentation zu den Produkten, die Sie kürzlich erworben haben.
Global Trade Item Number
| SKU | GTIN |
|---|---|
| M105-100MG | 04061833007549 |
| M105-10MG | 04061834037798 |
| M105-25MG | 04061834037811 |