Sélectionner une taille de conditionnement
| Conditionnement | Référence | Disponibilité | Prix |
|---|
A propos de cet article
biological source
synthetic (organic)
Quality Level
assay
≥98% (HPLC)
form
solid
color
yellow
mp
168-170 °C
solubility
H2O: slightly soluble 0.17 mg/mL, 45% (w/v) aq 2-hydroxypropyl-β-cyclodextrin: 2.8 mg/mL, 0.1 M HCl: slightly soluble, DMSO: soluble, aqueous buffer pH > 5: soluble, ethanol: soluble
storage temp.
2-8°C
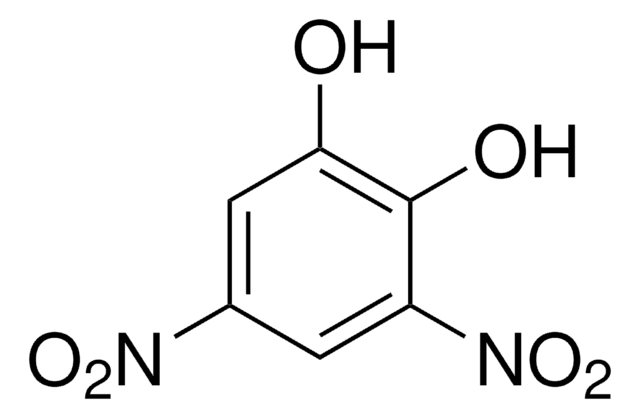
SMILES string
Oc1cc(cc(c1O)[N+]([O-])=O)[N+]([O-])=O
InChI
1S/C6H4N2O6/c9-5-2-3(7(11)12)1-4(6(5)10)8(13)14/h1-2,9-10H
InChI key
VDCDWNDTNSWDFJ-UHFFFAOYSA-N
Gene Information
human ... COMT(1312)
Application
Biochem/physiol Actions
Analysis Note
Disclaimer
1 of 1
Cet article | |||
|---|---|---|---|
| biological source synthetic (organic) | biological source - | biological source - | biological source synthetic (organic) |
| assay ≥98% (HPLC) | assay - | assay ≥98% | assay ≥98% (HPLC) |
| Gene Information human ... COMT(1312) | Gene Information human ... COMT(1312) | Gene Information human ... CACNA1C(775), CACNA1D(776), CACNA1F(778), CACNA1S(779) | Gene Information - |
| Quality Level 200 | Quality Level 100 | Quality Level 100 | Quality Level 200 |
| form solid | form solid | form powder | form powder or solid |
| solubility H2O: slightly soluble 0.17 mg/mL, 0.1 M HCl: slightly soluble, aqueous buffer pH > 5: soluble, 45% (w/v) aq 2-hydroxypropyl-β-cyclodextrin: 2.8 mg/mL, ethanol: soluble, DMSO: soluble | solubility H2O: slightly soluble <0.7 mg/mL, 45% (w/v) aq 2-hydroxypropyl-β-cyclodextrin: 1.0 mg/mL, ethanol: soluble | solubility DMSO: ~1 mg/mL, 0.1 M NaOH: insoluble, H2O: slightly soluble, methanol: soluble | solubility water: soluble 1 mg/mL |
Classe de stockage
11 - Combustible Solids
wgk
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
Eyeshields, Gloves, type N95 (US)
Faites votre choix parmi les versions les plus récentes :
Déjà en possession de ce produit ?
Retrouvez la documentation relative aux produits que vous avez récemment achetés dans la Bibliothèque de documents.
Numéro d'article de commerce international
| Référence | GTIN |
|---|---|
| 797391-200MG | 04061832939643 |
| D131-50MG | 04061832947129 |
| D131-250MG | 04061832947112 |