Fontos dokumentumok
66940
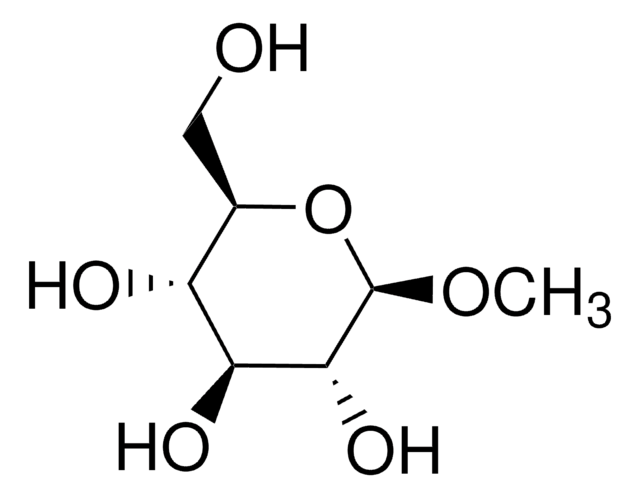
Methyl α-D-glucopyranoside
≥99.0%, suitable for microbiology
Szinonimák:
alpha-Methyl D-glucose ether, Methyl glucose, Methyl alpha-D-glucoside, Methyl glucopyranoside, Methyl alpha-D-glucose, Methyl pyranoside, Methyl α-D-glucoside
About This Item
Javasolt termékek
Minőségi szint
Teszt
≥99.0% (sum of enantiomers, HPLC)
≥99.0%
form
crystalline powder
optikai aktivitás
[α]20/D +157±3°, c = 10% in H2O
kiszerelés
pkg of 100 g
tárolási körülmény
(Keep container tightly closed in a dry and well-ventilated place.)
szín
colorless
mp
165-169 °C
alkalmazás(ok)
microbiology
SMILES string
CO[C@H]1O[C@H](CO)[C@@H](O)[C@H](O)[C@H]1O
InChI
1S/C7H14O6/c1-12-7-6(11)5(10)4(9)3(2-8)13-7/h3-11H,2H2,1H3/t3-,4-,5+,6-,7+/m1/s1
Nemzetközi kémiai azonosító kulcs
HOVAGTYPODGVJG-ZFYZTMLRSA-N
Looking for similar products? Látogasson el ide Útmutató a termékösszehasonlításhoz
Általános leírás
Alkalmazás
Tárolási osztály kódja
11 - Combustible Solids
WGK
WGK 1
Lobbanási pont (F)
Not applicable
Lobbanási pont (C)
Not applicable
Egyéni védőeszköz
Eyeshields, Gloves, type N95 (US)
Válasszon a legfrissebb verziók közül:
Már rendelkezik ezzel a termékkel?
Az Ön által nemrégiben megvásárolt termékekre vonatkozó dokumentumokat a Dokumentumtárban találja.
Az ügyfelek ezeket is megtekintették
Cikkek
Culture media provides a habitat with suitable nutrients, energy sources, and certain environmental conditions for the growth of microorganisms. The components of the culture media range from simple sugars to peptones, salts, antibiotics, and complex indicators.
Tudóscsoportunk valamennyi kutatási területen rendelkezik tapasztalattal, beleértve az élettudományt, az anyagtudományt, a kémiai szintézist, a kromatográfiát, az analitikát és még sok más területet.
Lépjen kapcsolatba a szaktanácsadással