Select a Size
| Pack Size | SKU | Availability | Price |
|---|
About This Item
grade
analytical standard
Quality Level
product line
PESTANAL®
shelf life
limited shelf life, expiry date on the label
technique(s)
HPLC: suitable, gas chromatography (GC): suitable
application(s)
agriculture
environmental
format
neat
storage temp.
2-8°C
SMILES string
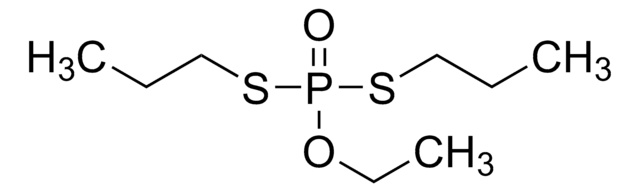
CCCSP(=O)(OCC)SCCC
InChI
1S/C8H19O2PS2/c1-4-7-12-11(9,10-6-3)13-8-5-2/h4-8H2,1-3H3
InChI key
VJYFKVYYMZPMAB-UHFFFAOYSA-N
Application
Legal Information
1 of 1
This Item | |||
|---|---|---|---|
| format neat | format - | format neat | format neat |
| technique(s) HPLC: suitable, gas chromatography (GC): suitable | technique(s) - | technique(s) HPLC: suitable, gas chromatography (GC): suitable | technique(s) HPLC: suitable, gas chromatography (GC): suitable |
| grade analytical standard | grade certified reference material, TraceCERT® | grade analytical standard | grade analytical standard |
| product line PESTANAL® | product line TraceCERT® | product line PESTANAL® | product line PESTANAL® |
| application(s) agriculture | application(s) - | application(s) agriculture | application(s) agriculture |
| storage temp. 2-8°C | storage temp. 2-8°C | storage temp. 2-8°C | storage temp. 2-8°C |
Still not finding the right product?
Explore all of our products under Ethoprophos
signalword
Danger
Hazard Classifications
Acute Tox. 1 Dermal - Acute Tox. 1 Inhalation - Acute Tox. 2 Oral - Aquatic Acute 1 - Aquatic Chronic 1 - Skin Sens. 1
Storage Class
6.1A - Combustible, acute toxic Cat. 1 and 2 / very toxic hazardous materials
wgk
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
Eyeshields, Faceshields, Gloves, type ABEK (EN14387) respirator filter
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Global Trade Item Number
| SKU | GTIN |
|---|---|
| 45306-100MG | 04061838474421 |