Sign In to View Organizational & Contract Pricing.
Select a Size
Change View
| Size/SKU | Availability | Price |
|---|
About This Item
Empirical Formula (Hill Notation):
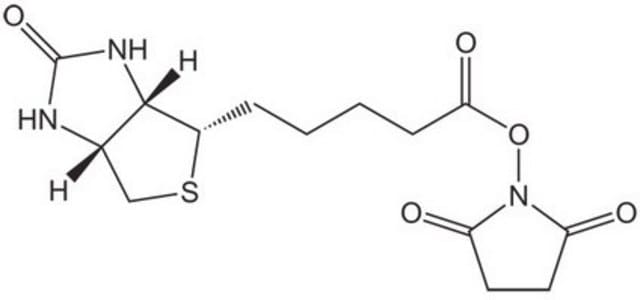
C18H31IN4O5S
Molecular Weight:
542.43
NACRES:
NA.32
PubChem Substance ID:
UNSPSC Code:
12352119
MDL number:
Form:
powder
Pricing and availability is not currently available.
description
Sulfhydryl specific, water soluble biotinylation reagent
Quality Segment
form
powder
solubility
H2O: 10 mg/mL
storage temp.
2-8°C
SMILES string
ICC(=O)NCCOCCOCCNC(=O)CCCCC1SCC2NC(=O)NC12
InChI
1S/C18H31IN4O5S/c19-11-16(25)21-6-8-28-10-9-27-7-5-20-15(24)4-2-1-3-14-17-13(12-29-14)22-18(26)23-17/h13-14,17H,1-12H2,(H,20,24)(H,21,25)(H2,22,23,26)
InChI key
LWGLKVINXNMNJX-UHFFFAOYSA-N
General description
Biotin polyethyleneoxide (PEO) iodoacetamide is a sulfhydryl (thiol) specific biotinylation reagent.[1]
Application
- Incorporates a 12 atom hydrophilic spacer.
- Typically coupled to sulfhydryl groups at pH 7.5 - 8.5
- Especially useful for cysteine labeling in many proteomics type applications such as peptide mapping and mass spectrometry.
Biotin Polyethyleneoxide Iodoacetamide has been used as a thiol reactive reagent to demonstrate the presence of free thiols in capsid protein L1 of human papillomavirus 16. It has been used for biotinylation of proteins for ELISA (enzyme-linked immunosorbent assay) competition and pull down experiments.
Biochem/physiol Actions
Biotin polyethyleneoxide (PEO) iodoacetamide group reacts specifically with reduced thiols at pH 7.5 - 8.5. Peptides and small molecules containing thiol groups may also be labeled using this reagent. The biotin PEO iodoacetamide enables tagging of cysteine residues and conjugation to sulfhydryls in proteins making it useful in proteomics, peptide mapping, phosphopeptide analysis, and mass spectrometry.
1 of 1
This Item | |||
|---|---|---|---|
| solubility H2O: 10 mg/mL | solubility - | solubility - | solubility - |
| Quality Level 200 | Quality Level 200 | Quality Level 200 | Quality Level 200 |
| storage temp. 2-8°C | storage temp. 15-25°C | storage temp. −20°C | storage temp. −20°C |
| form powder | form powder | form - | form - |
| description Sulfhydryl specific, water soluble biotinylation reagent | description - | description - | description - |
signalword
Warning
hcodes
Hazard Classifications
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
target_organs
Respiratory system
Storage Class
11 - Combustible Solids
wgk
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.