918350
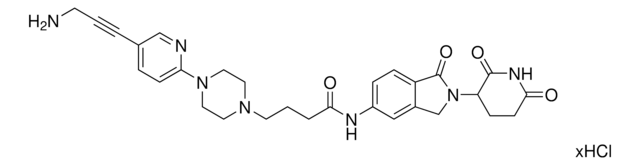
C5 Lenalidomide-alkyne-piperidine hydrochloride
≥95%
Synonim(y):
N-(2-(2,6-Dioxopiperidin-3-yl)-1-oxoisoindolin-5-yl)-3-(piperidin-4-yl)propiolamide hydrochloride, Crosslinker-E3 Ligase ligand conjugate, Protein degrader building block for PROTAC® research, Template for synthesis of targeted protein degrader
About This Item
Polecane produkty
ligand
C5 Lenalidomide
Próba
≥95%
Postać
powder or crystals
przydatność reakcji
reactivity: carboxyl reactive
reagent type: ligand-linker conjugate
grupa funkcyjna
amine
temp. przechowywania
2-8°C
ciąg SMILES
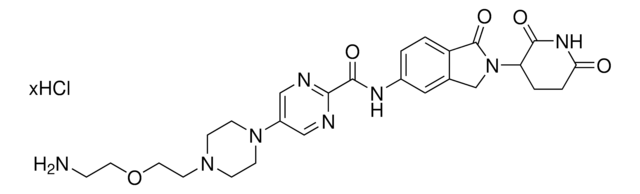
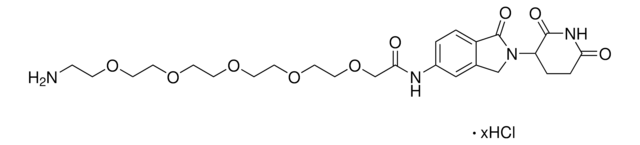
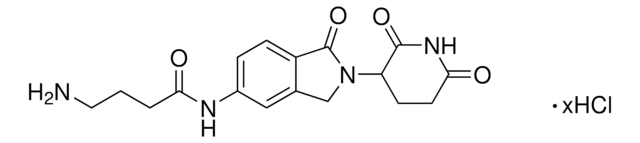
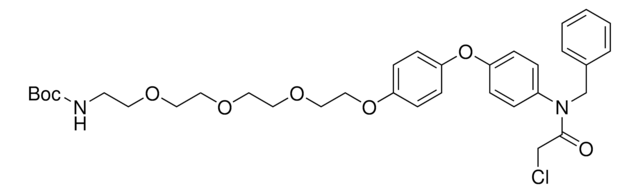
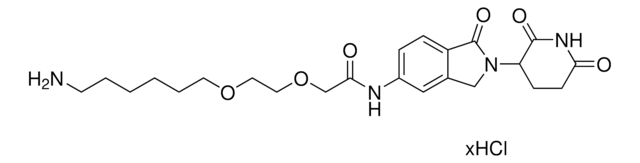
O=C1N(C2CCC(NC2=O)=O)CC3=CC(NC(C#CC4CCNCC4)=O)=CC=C31.Cl
Zastosowanie
Inne uwagi
Portal: Building PROTAC® Degraders for Targeted Protein Degradation
Targeted Protein Degradation by Small Molecules
Small-Molecule PROTACS: New Approaches to Protein Degradation
Targeted Protein Degradation: from Chemical Biology to Drug Discovery
Impact of linker length on the activity of PROTACs
Informacje prawne
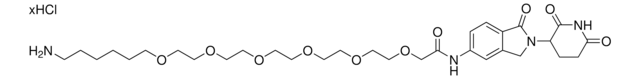
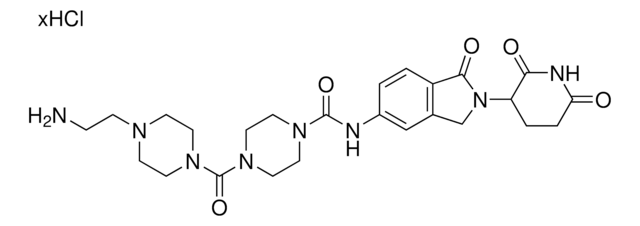
produkt powiązany
Kod klasy składowania
11 - Combustible Solids
Klasa zagrożenia wodnego (WGK)
WGK 3
Temperatura zapłonu (°F)
Not applicable
Temperatura zapłonu (°C)
Not applicable
Certyfikaty analizy (CoA)
Poszukaj Certyfikaty analizy (CoA), wpisując numer partii/serii produktów. Numery serii i partii można znaleźć na etykiecie produktu po słowach „seria” lub „partia”.
Masz już ten produkt?
Dokumenty związane z niedawno zakupionymi produktami zostały zamieszczone w Bibliotece dokumentów.
Produkty
Protein Degrader Building Blocks are a collection of crosslinker-E3 ligand conjugates with a pendant functional group for covalent linkage to a target ligand.
Nasz zespół naukowców ma doświadczenie we wszystkich obszarach badań, w tym w naukach przyrodniczych, materiałoznawstwie, syntezie chemicznej, chromatografii, analityce i wielu innych dziedzinach.
Skontaktuj się z zespołem ds. pomocy technicznej