Select a Size
| Pack Size | SKU | Availability | Price |
|---|
About This Item
biological source
Streptomyces sp.
Quality Level
assay
≥98% (HPLC)
form
powder
solubility
methanol: 10 mg/mL, clear, colorless
storage temp.
2-8°C
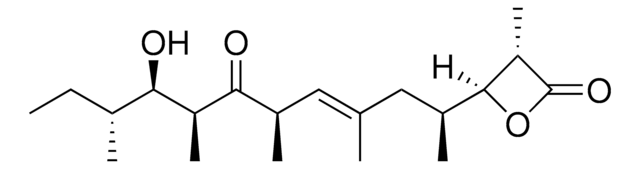
SMILES string
[H][C@@]1([C@H](C/C(C)=C/[C@H](C([C@H]([C@@H]([C@@H](CC)C)O)C)=O)C)C)OC([C@H]1C)=O
InChI
1S/C20H34O4/c1-8-12(3)17(21)15(6)18(22)13(4)9-11(2)10-14(5)19-16(7)20(23)24-19/h9,12-17,19,21H,8,10H2,1-7H3/b11-9+/t12-,13-,14+,15+,16+,17-,19+/m1/s1
InChI key
WOISDAHQBUYEAF-QIQXJRRPSA-N
General description
1 of 1
This Item | |||
|---|---|---|---|
| assay ≥98% (HPLC) | assay ≥99% (HPLC and TLC) | assay - | assay ≥98% (TLC) |
| biological source Streptomyces sp. | biological source Penicillium brefeldianum | biological source Penicillium brefeldianum | biological source Streptomyces sp. |
| Quality Level 100 | Quality Level 300 | Quality Level 200 | Quality Level 100 |
| form powder | form powder | form ready-to-use solution | form solid |
| solubility methanol: 10 mg/mL, clear, colorless | solubility DMSO: 10 mg/mL (Store stock solutions at -20 °C) | solubility DMSO: 10 mg/mL | solubility - |
| storage temp. 2-8°C | storage temp. 2-8°C | storage temp. 2-8°C | storage temp. −20°C |
Storage Class
11 - Combustible Solids
wgk
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
Eyeshields, Gloves, type N95 (US)
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Global Trade Item Number
| SKU | GTIN |
|---|---|
| E0761-1MG | 04061833601228 |