Fontos dokumentumok
SML2011
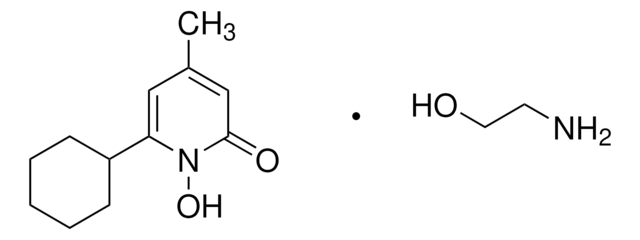
Ciclopirox
≥98% (HPLC)
Szinonimák:
Ciclopirox, 6-Cyclohexyl-1-hydroxy-4-methyl-2(1H)-pyridinone, HOE 296b, 6-Cyclohexyl-1-hydroxy-4-methyl-2(1H)-pyridone
About This Item
Javasolt termékek
Teszt
≥98% (HPLC)
Forma
powder
szín
white to beige
oldhatóság
DMSO: 2 mg/mL, clear
kiszállítva
ambient
tárolási hőmérséklet
2-8°C
SMILES string
ON1C(C2CCCCC2)=CC(C)=CC1=O
InChI
1S/C12H17NO2/c1-9-7-11(13(15)12(14)8-9)10-5-3-2-4-6-10/h7-8,10,15H,2-6H2,1H3
Nemzetközi kémiai azonosító kulcs
SCKYRAXSEDYPSA-UHFFFAOYSA-N
Looking for similar products? Látogasson el ide Útmutató a termékösszehasonlításhoz
Biokémiai/fiziológiai hatások
Figyelmeztetés
Warning
Figyelmeztető mondatok
Óvintézkedésre vonatkozó mondatok
Veszélyességi osztályok
Acute Tox. 4 Dermal - Acute Tox. 4 Inhalation - Acute Tox. 4 Oral - Eye Irrit. 2
Tárolási osztály kódja
11 - Combustible Solids
WGK
WGK 3
Válasszon a legfrissebb verziók közül:
Analitikai tanúsítványok (COA)
Nem találja a megfelelő verziót?
Ha egy adott verzióra van szüksége, a tétel- vagy cikkszám alapján rákereshet egy adott tanúsítványra.
Már rendelkezik ezzel a termékkel?
Az Ön által nemrégiben megvásárolt termékekre vonatkozó dokumentumokat a Dokumentumtárban találja.
Az ügyfelek ezeket is megtekintették
Tudóscsoportunk valamennyi kutatási területen rendelkezik tapasztalattal, beleértve az élettudományt, az anyagtudományt, a kémiai szintézist, a kromatográfiát, az analitikát és még sok más területet.
Lépjen kapcsolatba a szaktanácsadással