Sélectionner une taille de conditionnement
| Conditionnement | Référence | Disponibilité | Prix |
|---|
A propos de cet article
grade
pharmaceutical primary standard
API family
betamethasone
manufacturer/tradename
USP
application(s)
pharmaceutical (small molecule)
format
neat
storage temp.
2-8°C
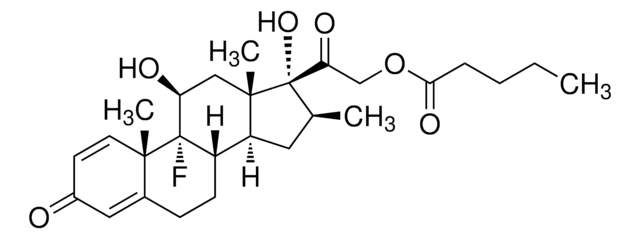
SMILES string
CCCCC(=O)OCC(=O)[C@@]1(O)[C@@H](C)C[C@H]2[C@@H]3CCC4=CC(=O)C=C[C@]4(C)[C@@]3(F)[C@@H](O)C[C@]12C
InChI
1S/C27H37FO6/c1-5-6-7-23(32)34-15-22(31)27(33)16(2)12-20-19-9-8-17-13-18(29)10-11-24(17,3)26(19,28)21(30)14-25(20,27)4/h10-11,13,16,19-21,30,33H,5-9,12,14-15H2,1-4H3/t16-,19-,20-,21-,24-,25-,26-,27-/m0/s1
InChI key
FEROCCAEIIKMJT-SUYDQAKGSA-N
General description
Application
- Betamethasone Valerate Cream
- Betamethasone Valerate Lotion
- Betamethasone Valerate Ointment
Analysis Note
Other Notes
1 of 1
Cet article | |||
|---|---|---|---|
| manufacturer/tradename USP | manufacturer/tradename USP | manufacturer/tradename - | manufacturer/tradename USP |
| grade pharmaceutical primary standard | grade pharmaceutical primary standard | grade certified reference material, pharmaceutical secondary standard | grade pharmaceutical primary standard |
| application(s) pharmaceutical (small molecule) | application(s) pharmaceutical (small molecule) | application(s) pharmaceutical | application(s) pharmaceutical (small molecule) |
| format neat | format neat | format - | format neat |
| storage temp. 2-8°C | storage temp. - | storage temp. 2-8°C | storage temp. - |
| API family betamethasone | API family betamethasone | API family - | API family betamethasone |
signalword
Danger
hcodes
Hazard Classifications
Repr. 1B - STOT RE 2
Classe de stockage
6.1C - Combustible acute toxic Cat.3 / toxic compounds or compounds which causing chronic effects
wgk
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
Faites votre choix parmi les versions les plus récentes :
Déjà en possession de ce produit ?
Retrouvez la documentation relative aux produits que vous avez récemment achetés dans la Bibliothèque de documents.
Numéro d'article de commerce international
| Référence | GTIN |
|---|---|
| 1069018-50MG | 04061833019771 |