Select a Size
| Pack Size | SKU | Availability | Price |
|---|
About This Item
Product Name
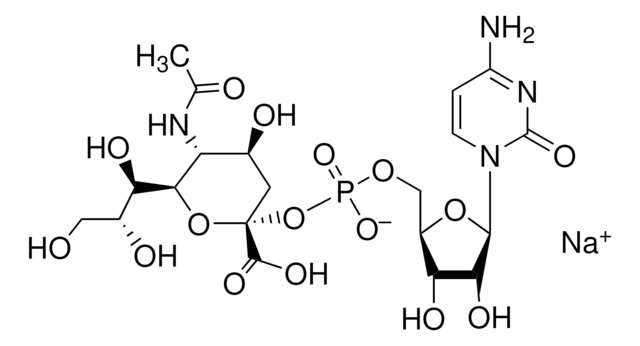
Cytidine-5′-monophospho-N-acetylneuraminic acid sodium salt, ≥85% (HPLC)
biological source
synthetic (organic)
Quality Level
assay
≥85% (HPLC)
form
powder
solubility
water: 50 mg/mL, clear, colorless
storage temp.
−20°C
SMILES string
[Na+].CC(=O)N[C@@H]1[C@@H](O)C[C@@](O[C@@H]1[C@H](O)[C@H](O)CO)(OP([O-])(=O)OC[C@H]2O[C@H]([C@H](O)[C@@H]2O)N3C=CC(N)=NC3=O)C(O)=O
InChI
1S/C20H31N4O16P.Na/c1-7(26)22-12-8(27)4-20(18(32)33,39-16(12)13(29)9(28)5-25)40-41(35,36)37-6-10-14(30)15(31)17(38-10)24-3-2-11(21)23-19(24)34;/h2-3,8-10,12-17,25,27-31H,4-6H2,1H3,(H,22,26)(H,32,33)(H,35,36)(H2,21,23,34);/q;+1/p-1/t8-,9+,10+,12+,13+,14+,15+,16?,17+,20+;/m0./s1
InChI key
VFRHSOGUONIUOR-CTFMUGKASA-M
General description
Application
- as a standard in high-performance anion-exchange chromatography with pulsed amperometric detection (HPAEC-PAD) for nucleotide sugar analysis in Joubert syndrome type 10 (JBTS10) patient cells and control skin fibroblasts[1],
- As a substrate for the enzymatic sialylation of G2 glycoforms[2], resialylation assay[3][4][5],
- in in-vitro sialyltransferase assay[6]
Biochem/physiol Actions
Preparation Note
CMP-NAN is very acid-labile.
1 of 1
This Item | |||
|---|---|---|---|
| biological source synthetic (organic) | biological source synthetic | biological source synthetic | biological source synthetic (organic) |
| Quality Level 200 | Quality Level 200 | Quality Level 100 | Quality Level 200 |
| assay ≥85% (HPLC) | assay ≥95% (HPLC) | assay ≥99.0% (HPLC) | assay ≥96% (HPLC) |
| form powder | form powder | form powder | form powder |
| storage temp. −20°C | storage temp. −20°C | storage temp. 2-8°C | storage temp. −20°C |
| solubility water: 50 mg/mL, clear, colorless | solubility water: 50 mg/mL, clear, colorless | solubility - | solubility H2O: 100 mg/mL |
Storage Class
11 - Combustible Solids
wgk
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Related Content
Glycobiology and Glycoproteomics Brochure
Global Trade Item Number
| SKU | GTIN |
|---|---|
| C8271-1MG | 04061833522011 |
| C8271-5MG | 04061833522035 |
| C8271-25MG | 04061833522028 |