Fontos dokumentumok
PHR1308
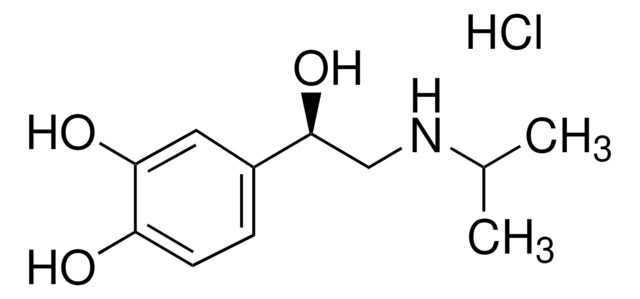
Propranolol hydrochloride
Pharmaceutical Secondary Standard; Certified Reference Material
Szinonimák:
(±)-Propranolol hydrochloride, (±)-1-Isopropylamino-3-(1-naphthyloxy)-2-propanol hydrochloride, DL-Propranolol hydrochloride
About This Item
Javasolt termékek
grade
certified reference material
pharmaceutical secondary standard
Minőségi szint
Ügynökség
traceable to Ph. Eur. P3500000
traceable to USP 1576005
API-család
propranolol
Analitikai műbizonylat
current certificate can be downloaded
technika/technikák
HPLC: suitable
gas chromatography (GC): suitable
alkalmazás(ok)
pharmaceutical (small molecule)
Formátum
neat
tárolási hőmérséklet
2-30°C
SMILES string
Cl.CC(C)NCC(O)COc1cccc2ccccc12
InChI
1S/C16H21NO2.ClH/c1-12(2)17-10-14(18)11-19-16-9-5-7-13-6-3-4-8-15(13)16;/h3-9,12,14,17-18H,10-11H2,1-2H3;1H
Nemzetközi kémiai azonosító kulcs
ZMRUPTIKESYGQW-UHFFFAOYSA-N
Géninformáció
human ... ADRB1(153) , ADRB2(154)
Looking for similar products? Látogasson el ide Útmutató a termékösszehasonlításhoz
Általános leírás
Propranolol is a β-adrenergic blocking compound, widely used for the management of cardiac arrhythmia, sinus tachycardia, angina pectoris and hypertension. It is also prescribed for the treatment of dysfunctional labor, migraine, as well as anxiety.
Alkalmazás
Analízis megjegyzés
Egyéb megjegyzések
Lábjegyzet
Javasolt termékek
kapcsolódó termék
Analitikai tanúsítványok (COA)
Analitikai tanúsítványok (COA) keresése a termék sarzs-/tételszámának megadásával. A sarzs- és tételszámok a termék címkéjén találhatók, a „Lot” vagy „Batch” szavak után.
Már rendelkezik ezzel a termékkel?
Az Ön által nemrégiben megvásárolt termékekre vonatkozó dokumentumokat a Dokumentumtárban találja.
Az ügyfelek ezeket is megtekintették
Tudóscsoportunk valamennyi kutatási területen rendelkezik tapasztalattal, beleértve az élettudományt, az anyagtudományt, a kémiai szintézist, a kromatográfiát, az analitikát és még sok más területet.
Lépjen kapcsolatba a szaktanácsadással