Fontos dokumentumok
Y0001380
Olanzapine for system suitability
European Pharmacopoeia (EP) Reference Standard
Szinonimák:
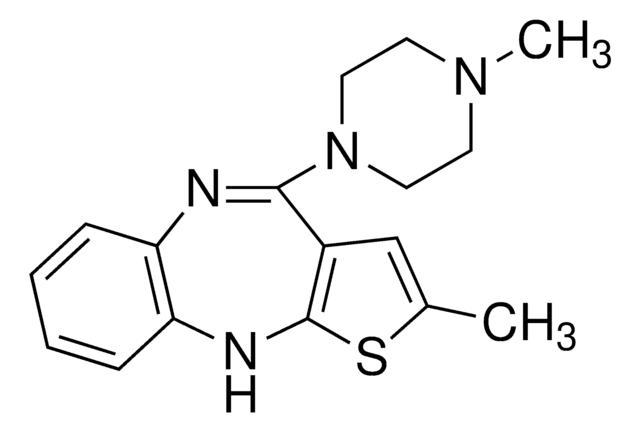
Olanzapine, 2-Methyl-4-(4-methyl-1-piperazinyl)- 10H-thieno[2,3-b][1,5]benzodiazepine
About This Item
Javasolt termékek
grade
pharmaceutical primary standard
API-család
olanzapine
gyártó/kereskedő neve
EDQM
alkalmazás(ok)
pharmaceutical (small molecule)
format
neat
tárolási hőmérséklet
2-8°C
SMILES string
CN1CCN(CC1)C2=Nc3ccccc3Nc4sc(C)cc24
InChI
1S/C17H20N4S/c1-12-11-13-16(21-9-7-20(2)8-10-21)18-14-5-3-4-6-15(14)19-17(13)22-12/h3-6,11,19H,7-10H2,1-2H3
Nemzetközi kémiai azonosító kulcs
KVWDHTXUZHCGIO-UHFFFAOYSA-N
Géninformáció
human ... DRD2(1813) , DRD3(1814) , DRD4(1815) , HTR2A(3356) , HTR2C(3358)
Looking for similar products? Látogasson el ide Útmutató a termékösszehasonlításhoz
Általános leírás
Alkalmazás
Kiszerelés
Egyéb megjegyzések
kapcsolódó termék
Figyelmeztetés
Danger
Figyelmeztető mondatok
Óvintézkedésre vonatkozó mondatok
Veszélyességi osztályok
Acute Tox. 3 Oral - Skin Irrit. 2 - Skin Sens. 1 - STOT SE 3
Célzott szervek
Central nervous system
Tárolási osztály kódja
6.1C - Combustible acute toxic Cat.3 / toxic compounds or compounds which causing chronic effects
WGK
WGK 3
Válasszon a legfrissebb verziók közül:
Analitikai tanúsítványok (COA)
Sajnos jelenleg COA nem áll rendelkezésre ehhez a termékhez online.
Ha segítségre van szüksége, lépjen velünk kapcsolatba Vevőszolgálat
Már rendelkezik ezzel a termékkel?
Az Ön által nemrégiben megvásárolt termékekre vonatkozó dokumentumokat a Dokumentumtárban találja.
Tudóscsoportunk valamennyi kutatási területen rendelkezik tapasztalattal, beleértve az élettudományt, az anyagtudományt, a kémiai szintézist, a kromatográfiát, az analitikát és még sok más területet.
Lépjen kapcsolatba a szaktanácsadással