MAB1562
Anti-Prion Protein Antibody, a.a. 109-112, clone 3F4
clone 3F4, Chemicon®, from mouse
Szinonimák:
PrP, CD230
About This Item
Javasolt termékek
biológiai forrás
mouse
Minőségi szint
antitest forma
purified immunoglobulin
antitest terméktípus
primary antibodies
klón
3F4, monoclonal
faj reaktivitás
hamster, human
gyártó/kereskedő neve
Chemicon®
technika/technikák
ELISA: suitable
immunohistochemistry (formalin-fixed, paraffin-embedded sections): suitable
immunoprecipitation (IP): suitable
western blot: suitable
izotípus
IgG2a
NCBI elérési szám
UniProt elérési szám
kiszállítva
dry ice
célzott transzláció utáni módosítás
unmodified
Géninformáció
human ... PRNP(5621)
Általános leírás
Egyediség
Immunogén
Alkalmazás
Representative images from a previous lot. Optimal Staining With Citrate Buffer, pH 6.0, Epitope Retrieval: Human Brain
Immunohistochemistry (Kitamoto et al., 1987):
1:100-1:1,000 *See protocol below.
Epitope must be re-exposed in fixed tissue by pretreatment of tissue using one of the following procedures:
a. formic acid for 10 minutes at room temperature (Kitamoto et al., 1987)
b. hydrolytic autoclaving (Kitamoto et al., 1991)
c. microwaving (BioGenex, San Ramon, CA)
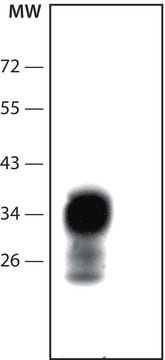
Western Blot: (Kascsak, R.J., 1991; Kascsak, R.J., 1987):
1:10,000-1:100,000 dilution of a previous lot was used.
Immunoprecipitation: (Kascsak, R.J., 1991; Kascsak, R.J., 1987):
1:10-1:100 dilution of a previous lot was used.
ELISA: (Kascsak, R.J., 1991; Kascsak, R.J., 1987):
1:100,000 dilution of a previous lot was used.
Optimal working dilutions must be determined by end user.
Minőség
Prion Protein (cat. # MAB1562) staining pattern/morphology in normal brain. Tissue was pretreated with Citrate, pH 6.0. This lot of antibody was diluted to 1:500, using IHC-Select Detection with HRP-DAB. Immunoreactivity is seen predominantly as cell body staining of neurons.
Optimal Staining With Citrate Buffer, pH 6.0, Epitope Retrieval: Human Brain
Cél megnevezése
Fizikai forma
Jogi információk
Nem találja a megfelelő terméket?
Próbálja ki a Termékválasztó eszköz. eszközt
Tárolási osztály kódja
12 - Non Combustible Liquids
WGK
WGK 2
Lobbanási pont (F)
Not applicable
Lobbanási pont (C)
Not applicable
Analitikai tanúsítványok (COA)
Analitikai tanúsítványok (COA) keresése a termék sarzs-/tételszámának megadásával. A sarzs- és tételszámok a termék címkéjén találhatók, a „Lot” vagy „Batch” szavak után.
Már rendelkezik ezzel a termékkel?
Az Ön által nemrégiben megvásárolt termékekre vonatkozó dokumentumokat a Dokumentumtárban találja.
Tudóscsoportunk valamennyi kutatási területen rendelkezik tapasztalattal, beleértve az élettudományt, az anyagtudományt, a kémiai szintézist, a kromatográfiát, az analitikát és még sok más területet.
Lépjen kapcsolatba a szaktanácsadással