M0600905
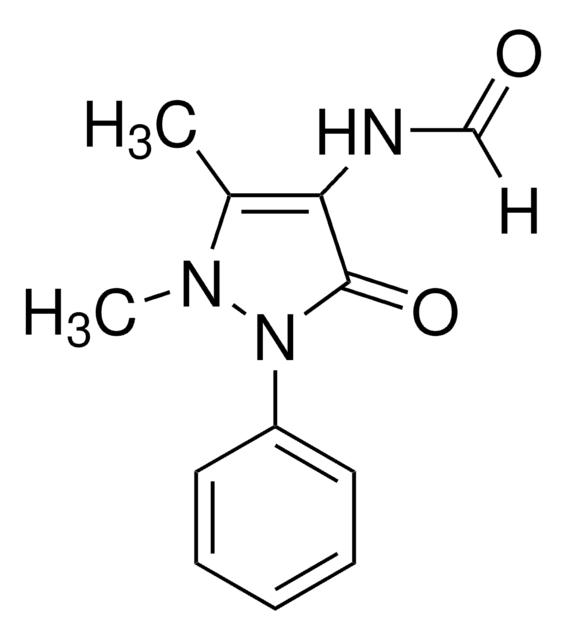
Metamizole impurity A
European Pharmacopoeia (EP) Reference Standard
別名:
N-(1,5-ジメチル-3-オキソ-2-フェニル-2,3-ジヒドロ-1H-ピラゾール-4-イル)ホルムアミド
ログイン組織・契約価格を表示する
すべての画像(1)
サイズを選択してください
表示を変更する
15 MG
¥19,900
About This Item
実験式(ヒル表記法):
C12H13N3O2
CAS番号:
分子量:
231.25
Beilstein:
217665
MDL番号:
UNSPSCコード:
41116107
PubChem Substance ID:
NACRES:
NA.24
おすすめの製品
グレード
pharmaceutical primary standard
APIファミリー
metamizole
メーカー/製品名
EDQM
アプリケーション
pharmaceutical (small molecule)
フォーマット
neat
SMILES記法
CN1N(C(=O)C(NC=O)=C1C)c2ccccc2
InChI
1S/C12H13N3O2/c1-9-11(13-8-16)12(17)15(14(9)2)10-6-4-3-5-7-10/h3-8H,1-2H3,(H,13,16)
InChI Key
WSJBSKRPKADYRQ-UHFFFAOYSA-N
類似した製品をお探しですか? 訪問 製品比較ガイド
詳細
この製品は薬局方標準品です。発行元の薬局方により製造・供給されています。MSDSを含む製品情報などの詳しい情報は、発行元の薬局方のウェブサイトよりご確認ください。
アプリケーション
Metamizole impurity A EP Reference standard, intended for use in laboratory tests only as specifically prescribed in the European Pharmacopoeia.
包装
The product is delivered as supplied by the issuing Pharmacopoeia. For the current unit quantity, please visit the EDQM reference substance catalogue.
その他情報
Sales restrictions may apply.
関連製品
製品番号
詳細
価格
シグナルワード
Warning
危険有害性情報
危険有害性の分類
Acute Tox. 4 Oral
保管分類コード
11 - Combustible Solids
WGK
WGK 3
引火点(°F)
Not applicable
引火点(℃)
Not applicable
最新バージョンのいずれかを選択してください:
この製品を見ている人はこちらもチェック
K Inoue et al.
Journal of chromatography, 274, 201-208 (1983-05-13)
Aminopyrine and its metabolites, including 3-hydroxymethyl-2-methyl-4-dimethylamino-1-phenyl-3-pyrazoline-5-one which is a hydroxylated metabolite of aminopyrine, were separated on a reversed-phase (C8) Radial-Pak column using a mobile phase of methanol-triethylamine-water (30:1:69) adjusted to pH 5.40 with acetic acid. Detection of the peak was
I Carretero et al.
The Analyst, 120(6), 1729-1732 (1995-06-01)
A rapid solid-phase extraction (SPE) procedure was developed for the quantitative isolation of three important antipyrine (dipyrone) metabolites from human plasma: 4-formylaminoantipyrine (FAA), 4-aminoantipyrine (AA) and 4-methylaminoantipyrine (MAA). Separation and quantitation were performed using micellar liquid chromatography (MLC) with a
S Imaoka et al.
Archives of biochemistry and biophysics, 265(1), 159-170 (1988-08-15)
Four aminopyrine metabolites generated by hepatic microsomes were simultaneously assayed by high-performance liquid chromatography. The metabolites were 4-monomethylaminoantipyrine (MAA), 4-aminoantipyrine (AA), 3-hydroxymethyl-2-methyl-4-dimethylamino-1-phenyl-3-pyrazoline-5-one (AM-OH), and one unidentified metabolite. MAA was the major metabolite generated by the microsomes; its formation was induced
K Matsuyama et al.
Journal of pharmacobio-dynamics, 6(11), 821-828 (1983-11-01)
The effect of phenobarbital (PB) and 3-methylcholanthrene (3-MC) on the metabolic behavior of aminopyrine (AM) was studied using an isolated hepatocyte system prepared from male Wistar rats. The formation of 4-formylaminoantipyrine (FAA) was increased after pretreatment with PB, but not
M Levy et al.
European journal of clinical pharmacology, 57(6-7), 461-465 (2001-11-09)
We previously found that, compared with healthy subjects. asymptomatic hepatitis-B virus (HBV) carriers displaying slow acetylator phenotype demonstrate a significant prolongation of the elimination half-life of 4-methylaminoantipyrine (MAA) and a decrease in the clearance of formation of 4-aminoantipyrine (AA) and
アクティブなフィルタ
ライフサイエンス、有機合成、材料科学、クロマトグラフィー、分析など、あらゆる分野の研究に経験のあるメンバーがおります。.
製品に関するお問い合わせはこちら(テクニカルサービス)