Fontos dokumentumok
PHR1023
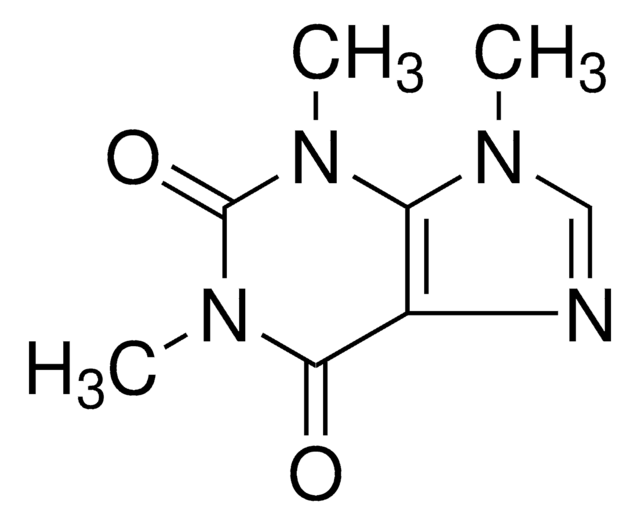
Theophylline
Pharmaceutical Secondary Standard; Certified Reference Material
Szinonimák:
1,3-Dimethylxanthine, 2,6-Dihydroxy-1,3-dimethylpurine, 3,7-Dihydro-1,3-dimethyl-1H-purine-2,6-dione
About This Item
Javasolt termékek
grade
certified reference material
pharmaceutical secondary standard
Minőségi szint
Ügynökség
traceable to BP 327
traceable to Ph. Eur. T080000
traceable to USP 1653004
API-család
theophylline
Analitikai műbizonylat
current certificate can be downloaded
technika/technikák
HPLC: suitable
gas chromatography (GC): suitable
alkalmazás(ok)
food and beverages
pharmaceutical (small molecule)
Formátum
neat
tárolási hőmérséklet
2-30°C
SMILES string
CN1C(=O)N(C)c2[nH]cnc2C1=O
InChI
1S/C7H8N4O2/c1-10-5-4(8-3-9-5)6(12)11(2)7(10)13/h3H,1-2H3,(H,8,9)
Nemzetközi kémiai azonosító kulcs
ZFXYFBGIUFBOJW-UHFFFAOYSA-N
Géninformáció
human ... ADORA1(134) , ADORA2A(135) , ADORA2B(136) , ADORA3(140) , PDE3A(5139) , PDE3B(5140) , PDE4A(5141) , PDE4B(5142) , PDE4C(5143) , PDE4D(5144)
Looking for similar products? Látogasson el ide Útmutató a termékösszehasonlításhoz
Általános leírás
Alkalmazás
Biokémiai/fiziológiai hatások
Analízis megjegyzés
Egyéb megjegyzések
Lábjegyzet
Javasolt termékek
kapcsolódó termék
Figyelmeztetés
Danger
Figyelmeztető mondatok
Óvintézkedésre vonatkozó mondatok
Veszélyességi osztályok
Acute Tox. 3 Oral - Repr. 1B
Tárolási osztály kódja
6.1C - Combustible acute toxic Cat.3 / toxic compounds or compounds which causing chronic effects
WGK
WGK 1
Lobbanási pont (F)
Not applicable
Lobbanási pont (C)
Not applicable
Analitikai tanúsítványok (COA)
Analitikai tanúsítványok (COA) keresése a termék sarzs-/tételszámának megadásával. A sarzs- és tételszámok a termék címkéjén találhatók, a „Lot” vagy „Batch” szavak után.
Már rendelkezik ezzel a termékkel?
Az Ön által nemrégiben megvásárolt termékekre vonatkozó dokumentumokat a Dokumentumtárban találja.
Az ügyfelek ezeket is megtekintették
Tudóscsoportunk valamennyi kutatási területen rendelkezik tapasztalattal, beleértve az élettudományt, az anyagtudományt, a kémiai szintézist, a kromatográfiát, az analitikát és még sok más területet.
Lépjen kapcsolatba a szaktanácsadással