1A00800
USP
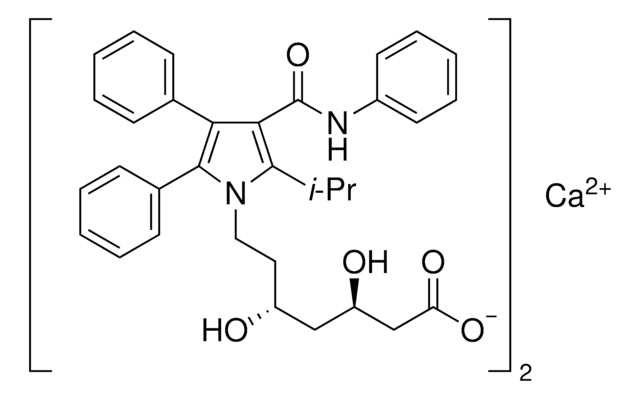
ATORVASTATIN EPOXY TETRAHYDROFURAN ANALOG
Pharmaceutical Analytical Impurity (PAI)
Szinonimák:
4-(4-Fluorophenyl)-2,4-dihydroxy-2-(1-methylethyl)-N,5-diphenyl-3,6-dioxabicyclo[3.1.0]hexane-1-carboxamide
Bejelentkezésa Szervezeti és Szerződéses árazás megtekintéséhez
Összes fotó(1)
About This Item
UNSPSC kód:
41116107
NACRES:
NA.24
Javasolt termékek
grade
pharmaceutical analytical impurity (PAI)
Ügynökség
USP
API-család
atorvastatin
gyártó/kereskedő neve
USP
alkalmazás(ok)
pharmaceutical
format
neat
tárolási hőmérséklet
2-8°C
Általános leírás
ATORVASTATIN EPOXY TETRAHYDROFURAN ANALOG is a USP Pharmaceutical Analytical Impurity (PAI).
USP PAI are a product line of impurities suitable for research and analytical purposes, which help to ensure the quality and safety of medicines.
Associated Drug Substance: Atorvastatin Calcium
Therapeutic Area: Antihyperlipidemics
For more information about this PAI, visit here.
USP PAI are a product line of impurities suitable for research and analytical purposes, which help to ensure the quality and safety of medicines.
Associated Drug Substance: Atorvastatin Calcium
Therapeutic Area: Antihyperlipidemics
For more information about this PAI, visit here.
Alkalmazás
ATORVASTATIN EPOXY TETRAHYDROFURAN ANALOG (USP PAI) is intended for use in analytical testing to detect, identify, and measure pharmaceutical impurities.
Tulajdonságok és előnyök
USP PAI advance your early analytical R&D and process development. PAI can be used in the following applications:
1. Conduct analytical tests during early formulation feasibility studies.
2. Determine degradation impurities produced during stress studies.
3. Develop, validate, and transfer analytical methods.
4. Perform spiking studies during process R&D to demonstrate depletion upon recrystallization.
5. Record retention times and/or spectra
6. Determine relative response factors.
7. Identify unknown impurities that formed during ICH stability conditions.
8. Identify impurities that are present in the Reference Listed Drug
9. Test for and profile impurities not listed in drug substance and drug product monographs.
1. Conduct analytical tests during early formulation feasibility studies.
2. Determine degradation impurities produced during stress studies.
3. Develop, validate, and transfer analytical methods.
4. Perform spiking studies during process R&D to demonstrate depletion upon recrystallization.
5. Record retention times and/or spectra
6. Determine relative response factors.
7. Identify unknown impurities that formed during ICH stability conditions.
8. Identify impurities that are present in the Reference Listed Drug
9. Test for and profile impurities not listed in drug substance and drug product monographs.
Analízis megjegyzés
These products are for test and assay use only. They are not meant for administration to humans or animals and cannot be used to diagnose, treat, or cure diseases of any kind.
Egyéb megjegyzések
Sales restrictions may apply.
Tárolási osztály kódja
11 - Combustible Solids
WGK
WGK 3
Lobbanási pont (F)
Not applicable
Lobbanási pont (C)
Not applicable
Analitikai tanúsítványok (COA)
Analitikai tanúsítványok (COA) keresése a termék sarzs-/tételszámának megadásával. A sarzs- és tételszámok a termék címkéjén találhatók, a „Lot” vagy „Batch” szavak után.
Már rendelkezik ezzel a termékkel?
Az Ön által nemrégiben megvásárolt termékekre vonatkozó dokumentumokat a Dokumentumtárban találja.
Tudóscsoportunk valamennyi kutatási területen rendelkezik tapasztalattal, beleértve az élettudományt, az anyagtudományt, a kémiai szintézist, a kromatográfiát, az analitikát és még sok más területet.
Lépjen kapcsolatba a szaktanácsadással