Összes fotó(1)
Fontos dokumentumok
Összes fotó(1)
About This Item
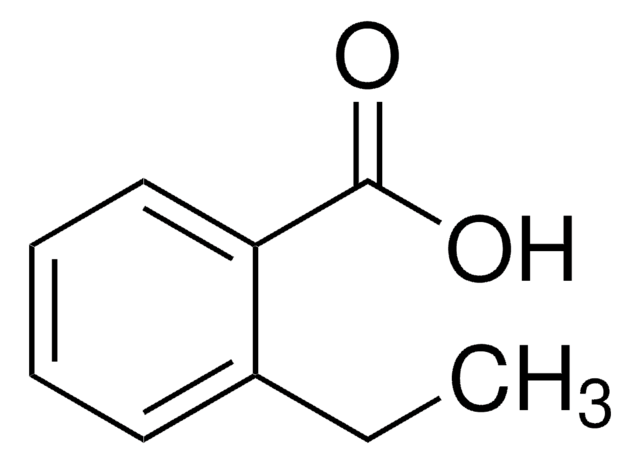
Lineáris képlet:
C2H5C6H4CO2H
CAS-szám:
Molekulatömeg:
150.17
Beilstein:
2041840
EC-szám:
MDL-szám:
UNSPSC kód:
12352100
PubChem Substance ID:
NACRES:
NA.22
Javasolt termékek
Minőségi szint
Teszt
99%
Forma
solid
mp
112-113 °C (lit.)
funkcionális csoport
carboxylic acid
SMILES string
CCc1ccc(cc1)C(O)=O
InChI
1S/C9H10O2/c1-2-7-3-5-8(6-4-7)9(10)11/h3-6H,2H2,1H3,(H,10,11)
Nemzetközi kémiai azonosító kulcs
ZQVKTHRQIXSMGY-UHFFFAOYSA-N
Looking for similar products? Látogasson el ide Útmutató a termékösszehasonlításhoz
Általános leírás
4-Ethylbenzoic acid reacts with lanthanum nitrate in aqueous solution to yield the polymer catena-poly[[aqua(4-ethylbenzoic acid-κO)lanthanum(III)]-tri-μ-4-ethylbenzoato].
Alkalmazás
4-Ethylbenzoic acid was used in the synthesis of ethyl 4-vinyl-α-cyano-β-phenylcinnamate. It was also used to functionalize the edge of “pristine” graphite in the presence of polyphosphoric acid/phosphorus pentoxide.
Tárolási osztály kódja
11 - Combustible Solids
WGK
WGK 3
Lobbanási pont (F)
Not applicable
Lobbanási pont (C)
Not applicable
Egyéni védőeszköz
Eyeshields, Gloves, type N95 (US)
Válasszon a legfrissebb verziók közül:
Már rendelkezik ezzel a termékkel?
Az Ön által nemrégiben megvásárolt termékekre vonatkozó dokumentumokat a Dokumentumtárban találja.
Az ügyfelek ezeket is megtekintették
Juan Yang et al.
Acta crystallographica. Section E, Structure reports online, 66(Pt 2), m183-m184 (2010-01-01)
The reaction of lanthanum nitrate and 4-ethyl-benzoic acid (EBAH) in aqueous solution yielded the title polymer, [La(C(9)H(9)O(2))(3)(C(9)H(10)O(2))(H(2)O)](n). The asymmetric unit contains one La(III) atom, three 4-ethyl-benzoate (EBA) ligands, one neutral EBAH ligand and one coordinated water mol-ecule. Each La(III) ion
Functional Polymers. VII. Ethyl 4-Vinyl-α-cyano-β-phenylcinnamate.
Sumida Y and Vogl O.
Polymer Journal, 13(6), 521-536 (1981)
Ruthenium(II)-catalyzed synthesis of hydroxylated arenes with ester as an effective directing group.
Yiqing Yang et al.
Organic letters, 14(11), 2874-2877 (2012-05-16)
An unprecedented Ru(II) catalyzed ortho-hydroxylation has been developed for the facile synthesis of a variety of multifunctionalized arenes from easily accessible ethyl benzoates with ester as an efficient directing group. Both the TFA/TFAA cosolvent system and oxidants serve as the
José María Moreno et al.
Chemical science, 10(7), 2053-2066 (2019-03-08)
Novel MOF-type materials with different morphologies based on assembled 1D organic-inorganic sub-domains were prepared using specific monodentate benzylcarboxylate spacers with functional substituents in the para-position as structure modulating agents. The combination of electron-withdrawing or electron-donating functions in the organic spacers
D M Jung et al.
Journal of agricultural and food chemistry, 48(2), 407-412 (2000-02-26)
Changes in flavor release and aroma characteristics in a medium including food phenolics may be attributed to an intermolecular interaction between flavor compounds and phenolics. To investigate the interaction, one- and two-dimensional NMR studies have been carried out on the
Tudóscsoportunk valamennyi kutatási területen rendelkezik tapasztalattal, beleértve az élettudományt, az anyagtudományt, a kémiai szintézist, a kromatográfiát, az analitikát és még sok más területet.
Lépjen kapcsolatba a szaktanácsadással