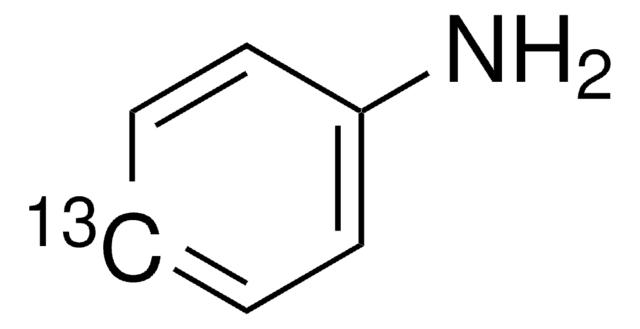
491799
Aniline-1-13C
99 atom % 13C
Synonyma:
Benzenamine-1-13C
About This Item
Doporučené produkty
isotopic purity
99 atom % 13C
assay
99% (CP)
refractive index
n20/D 1.586 (lit.)
bp
184 °C (lit.)
mp
−6 °C (lit.)
density
1.033 g/mL at 25 °C
mass shift
M+1
storage temp.
2-8°C
SMILES string
N[13c]1ccccc1
InChI
1S/C6H7N/c7-6-4-2-1-3-5-6/h1-5H,7H2/i6+1
InChI key
PAYRUJLWNCNPSJ-PTQBSOBMSA-N
Související kategorie
Packaging
signalword
Danger
Hazard Classifications
Acute Tox. 3 Dermal - Acute Tox. 3 Inhalation - Acute Tox. 3 Oral - Aquatic Acute 1 - Aquatic Chronic 1 - Carc. 2 - Eye Dam. 1 - Muta. 2 - Resp. Sens. 1
Storage Class
6.1A - Combustible acute toxic Cat. 1 and 2 / very toxic hazardous materials
wgk_germany
WGK 3
flash_point_f
158.0 °F - closed cup
flash_point_c
70 °C - closed cup
Osvědčení o analýze (COA)
Vyhledejte osvědčení Osvědčení o analýze (COA) zadáním čísla šarže/dávky těchto produktů. Čísla šarže a dávky lze nalézt na štítku produktu za slovy „Lot“ nebo „Batch“.
Již tento produkt vlastníte?
Dokumenty související s produkty, které jste v minulosti zakoupili, byly za účelem usnadnění shromážděny ve vaší Knihovně dokumentů.
Náš tým vědeckých pracovníků má zkušenosti ve všech oblastech výzkumu, včetně přírodních věd, materiálových věd, chemické syntézy, chromatografie, analytiky a mnoha dalších..
Obraťte se na technický servis.