H6012
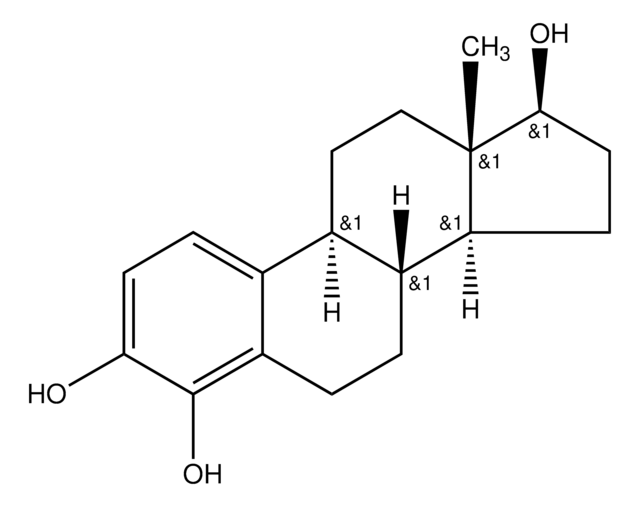
4-Hydroxyestrone
≥90% (HPLC)
Sinônimo(s):
1,3,5(10)-Estratriene-3,4-diol-17-one
About This Item
Produtos recomendados
esterilidade
non-sterile
Ensaio
≥90% (HPLC)
forma
powder
solubilidade
methanol: 10 mg/mL, clear, colorless to faintly brownish-yellow
Condições de expedição
ambient
temperatura de armazenamento
−20°C
cadeia de caracteres SMILES
[H][C@]12CC[C@]3(C)C(=O)CC[C@@]3([H])[C@]1([H])CCc4c(O)c(O)ccc24
InChI
1S/C18H22O3/c1-18-9-8-11-10-4-6-15(19)17(21)13(10)3-2-12(11)14(18)5-7-16(18)20/h4,6,11-12,14,19,21H,2-3,5,7-9H2,1H3/t11-,12-,14+,18+/m1/s1
chave InChI
XQZVQQZZOVBNLU-QDTBLXIISA-N
Ações bioquímicas/fisiológicas
Código de classe de armazenamento
11 - Combustible Solids
Classe de risco de água (WGK)
WGK 3
Ponto de fulgor (°F)
199.9 °F - closed cup
Ponto de fulgor (°C)
93.3 °C - closed cup
Equipamento de proteção individual
Eyeshields, Gloves, type N95 (US)
Certificados de análise (COA)
Busque Certificados de análise (COA) digitando o Número do Lote do produto. Os números de lote e remessa podem ser encontrados no rótulo de um produto após a palavra “Lot” ou “Batch”.
Já possui este produto?
Encontre a documentação dos produtos que você adquiriu recentemente na biblioteca de documentos.
Nossa equipe de cientistas tem experiência em todas as áreas de pesquisa, incluindo Life Sciences, ciência de materiais, síntese química, cromatografia, química analítica e muitas outras.
Entre em contato com a assistência técnica