所有图片(1)
About This Item
经验公式(希尔记法):
C14H20O10
CAS号:
分子量:
348.30
EC號碼:
MDL號碼:
分類程式碼代碼:
12352201
PubChem物質ID:
NACRES:
NA.22
推荐产品
品質等級
化驗
98%
形狀
solid
光學活性
[α]20/D +11°, c = 6 in chloroform
mp
126-128 °C (lit.)
SMILES 字串
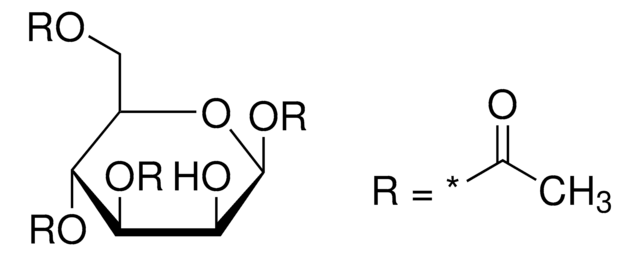
CC(=O)O[C@@H]1O[C@H](CO)[C@@H](OC(C)=O)[C@H](OC(C)=O)[C@H]1OC(C)=O
InChI
1S/C14H20O10/c1-6(16)20-11-10(5-15)24-14(23-9(4)19)13(22-8(3)18)12(11)21-7(2)17/h10-15H,5H2,1-4H3/t10-,11-,12+,13-,14-/m1/s1
InChI 密鑰
FEQXFAYSNRWXDW-RKQHYHRCSA-N
一般說明
1,2,3,4-Tetra-O-acetyl-β-ᴅ-glucopyranoseis a carbohydrate that is used in the synthesis of disaccharides and D-glucose6-phosphate.
應用
经证实,该磷酰化衍生物在肌醇合成酶的底物研究以及阴离子表面活性剂的制备中很有价值。
儲存類別代碼
11 - Combustible Solids
水污染物質分類(WGK)
WGK 3
閃點(°F)
Not applicable
閃點(°C)
Not applicable
個人防護裝備
Eyeshields, Gloves, type N95 (US)
其他客户在看
Beata Liberek et al.
Carbohydrate research, 341(13), 2275-2285 (2006-07-15)
The single-crystal X-ray diffraction and high-resolution 1H and 13C NMR spectral data for methyl 2,5-di-O-acetyl-beta-D-glucofuranosidurono-6,3-lactone and 1,2,5-tri-O-acetyl-beta-D-glucofuranurono-6,3-lactone are reported. The lactones were synthesized as byproducts of reactions carried out to obtain methyl 1,2,3,4-tetra-O-acetyl-D-glucopyranuronate. The conformations of these lactones in the
Yuriko Y Root et al.
Carbohydrate research, 337(21-23), 2343-2346 (2002-11-16)
The identity of the crystalline product formed by the acetylation of a mixture of methyl alpha- and beta-D-glucopyranuronates has been confirmed as being methyl 1,2,3,4-tetra-O-acetyl-beta-D-glucopyranuronate (3), which agrees with the assignment from 1H NMR. The absolute configuration of compound 3
A Milius et al.
Carbohydrate research, 229(2), 323-336 (1992-05-22)
D-Glucose 3- and 6-[sodium 2-(perfluoro-hexyl or -octyl)ethyl phosphates) have been synthesized by condensation of 1,2,3,4,-tetra-O-acetyl-beta-D-glucopyranose and 1,2:5,6-di-O-isopropylidene-alpha-D-glucofuranose with 2-(perfluoroalkyl)ethylphosphoroditriazolides followed by O-deacetylation or deacetalation. The structures of the compounds were established on the basis of 1H-, 19F-, 31P-, and 13C-NMR
Tetrahedron, 47, 3895-3895 (1991)
Wei Wang et al.
Glycoconjugate journal, 31(2), 133-143 (2013-11-13)
A group of fluorescent statistical glycopolymers, prepared via reversible addition-fragmentation chain-transfer (RAFT)-based polymerizations, were successfully employed in lectin-mediated bacterial binding studies. The resultant glycopolymers contained three different monomers: N-(2-hydroxyethyl) acrylamide (HEAA), N-(2-aminoethyl) methacrylamide (AEMA) and N-(2-glyconamidoethyl)-methacrylamides possessing different pendant sugars.
我们的科学家团队拥有各种研究领域经验,包括生命科学、材料科学、化学合成、色谱、分析及许多其他领域.
联系技术服务部门











![4,7,13,16,21,24-六氧-1,10-二氮双环[8.8.8]二十六烷 98%](/deepweb/assets/sigmaaldrich/product/structures/189/812/8a6555e5-8de6-4236-865f-19339cee3634/640/8a6555e5-8de6-4236-865f-19339cee3634.png)
