MAB1562
Anti-Prion Protein Antibody, a.a. 109-112, clone 3F4
clone 3F4, Chemicon®, from mouse
Synonim(y):
PrP, CD230
Wybierz wielkość
2290,00 zł
Przewidywany termin wysyłki25 kwietnia 2025Szczegóły
Wybierz wielkość
About This Item
2290,00 zł
Przewidywany termin wysyłki25 kwietnia 2025Szczegóły
Polecane produkty
pochodzenie biologiczne
mouse
Poziom jakości
forma przeciwciała
purified immunoglobulin
rodzaj przeciwciała
primary antibodies
klon
3F4, monoclonal
reaktywność gatunkowa
hamster, human
producent / nazwa handlowa
Chemicon®
metody
ELISA: suitable
immunohistochemistry (formalin-fixed, paraffin-embedded sections): suitable
immunoprecipitation (IP): suitable
western blot: suitable
izotyp
IgG2a
numer dostępu NCBI
numer dostępu UniProt
Warunki transportu
dry ice
docelowa modyfikacja potranslacyjna
unmodified
informacje o genach
human ... PRNP(5621)
Opis ogólny
Specyficzność
Immunogen
Zastosowanie
Representative images from a previous lot. Optimal Staining With Citrate Buffer, pH 6.0, Epitope Retrieval: Human Brain
Immunohistochemistry (Kitamoto et al., 1987):
1:100-1:1,000 *See protocol below.
Epitope must be re-exposed in fixed tissue by pretreatment of tissue using one of the following procedures:
a. formic acid for 10 minutes at room temperature (Kitamoto et al., 1987)
b. hydrolytic autoclaving (Kitamoto et al., 1991)
c. microwaving (BioGenex, San Ramon, CA)
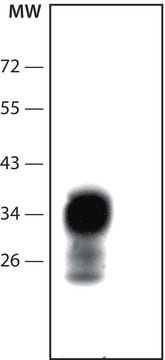
Western Blot: (Kascsak, R.J., 1991; Kascsak, R.J., 1987):
1:10,000-1:100,000 dilution of a previous lot was used.
Immunoprecipitation: (Kascsak, R.J., 1991; Kascsak, R.J., 1987):
1:10-1:100 dilution of a previous lot was used.
ELISA: (Kascsak, R.J., 1991; Kascsak, R.J., 1987):
1:100,000 dilution of a previous lot was used.
Optimal working dilutions must be determined by end user.
Jakość
Prion Protein (cat. # MAB1562) staining pattern/morphology in normal brain. Tissue was pretreated with Citrate, pH 6.0. This lot of antibody was diluted to 1:500, using IHC-Select Detection with HRP-DAB. Immunoreactivity is seen predominantly as cell body staining of neurons.
Optimal Staining With Citrate Buffer, pH 6.0, Epitope Retrieval: Human Brain
Opis wartości docelowych
Postać fizyczna
Informacje prawne
Nie możesz znaleźć właściwego produktu?
Wypróbuj nasz Narzędzie selektora produktów.
Kod klasy składowania
12 - Non Combustible Liquids
Klasa zagrożenia wodnego (WGK)
WGK 2
Temperatura zapłonu (°F)
Not applicable
Temperatura zapłonu (°C)
Not applicable
Certyfikaty analizy (CoA)
Poszukaj Certyfikaty analizy (CoA), wpisując numer partii/serii produktów. Numery serii i partii można znaleźć na etykiecie produktu po słowach „seria” lub „partia”.
Masz już ten produkt?
Dokumenty związane z niedawno zakupionymi produktami zostały zamieszczone w Bibliotece dokumentów.
Active Filters
Nasz zespół naukowców ma doświadczenie we wszystkich obszarach badań, w tym w naukach przyrodniczych, materiałoznawstwie, syntezie chemicznej, chromatografii, analityce i wielu innych dziedzinach.
Skontaktuj się z zespołem ds. pomocy technicznej