809160
N-Ethyl-N-isopropylpropan-2-aminium 3-(4-Acetyl-2,3,5,6-tetrafluorophenyl)-4-oxo-1,5-dioxaspiro[5.5]-undec-2-en-2-olate
About This Item
Prodotti consigliati
Stato
powder
Temperatura di conservazione
2-8°C
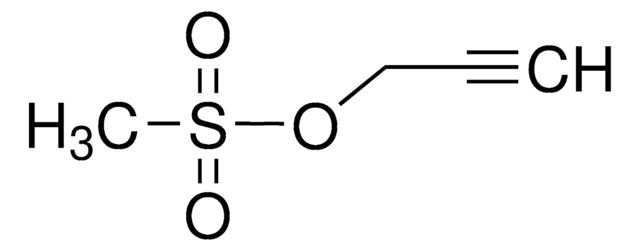
Stringa SMILE
FC1=C(C(C)=O)C(F)=C(F)C(C2=C([O-])OC3(CCCCC3)OC2=O)=C1F.CC[NH+](C(C)C)C(C)C
InChI
1S/C17H14F4O5.C8H19N/c1-7(22)8-11(18)13(20)9(14(21)12(8)19)10-15(23)25-17(26-16(10)24)5-3-2-4-6-17;1-6-9(7(2)3)8(4)5/h23H,2-6H2,1H3;7-8H,6H2,1-5H3
UYTIIFFVFGALQP-UHFFFAOYSA-N
Descrizione generale
Applicazioni
Scegli una delle versioni più recenti:
Certificati d'analisi (COA)
It looks like we've run into a problem, but you can still download Certificates of Analysis from our Documenti section.
Se ti serve aiuto, non esitare a contattarci Servizio Clienti
Possiedi già questo prodotto?
I documenti relativi ai prodotti acquistati recentemente sono disponibili nell’Archivio dei documenti.
Contenuto correlato
Organofluorine chemistry is an essential part of drug discovery programs as well as agrochemical programs and even plays a major role in materials chemistry. Despite the undeniable importance of fluorinated organic molecules, our ability to synthesize these substrates is lacking - though arguably it is better than that of Nature. Consequently, methods that allow facile access to fluorinated molecules are important especially when they provide unique access to fluorinated chemical space.
Filtri attivi
Il team dei nostri ricercatori vanta grande esperienza in tutte le aree della ricerca quali Life Science, scienza dei materiali, sintesi chimica, cromatografia, discipline analitiche, ecc..
Contatta l'Assistenza Tecnica.