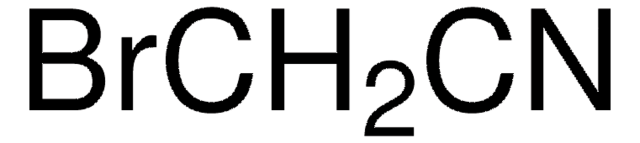766917
Lithium bis(triméthylsilyl)amide solution
1.5 M in THF
Synonyme(s) :
Hexaméthyldisilazane lithium salt
About This Item
Produits recommandés
Forme
liquid
Niveau de qualité
Concentration
1.5 M in THF
Densité
0.893 g/mL at 25 °C
Chaîne SMILES
[Li]N([Si](C)(C)C)[Si](C)(C)C
InChI
1S/C6H18NSi2.Li/c1-8(2,3)7-9(4,5)6;/h1-6H3;/q-1;+1
Clé InChI
YNESATAKKCNGOF-UHFFFAOYSA-N
Vous recherchez des produits similaires ? Visite Guide de comparaison des produits
Catégories apparentées
Description générale
Application
- In the deprotonation and nucleophilic difluoromethylation reactions.
- 3-methoxy substituted dihydropyrrole derivatives by reacting with aldehydes and lithiated methoxyallene via in situ formations of N-trimethylsilylated imines.
- In Darzens condensation and directed aldol condensation reactions.
- To synthesize poly(N-octyl-p-benzamide)s by chain-growth polycondensation of 4-octylaminobenzoic acid methyl ester.
Mention d'avertissement
Danger
Mentions de danger
Classification des risques
Carc. 2 - Eye Dam. 1 - Flam. Liq. 2 - Self-heat. 1 - Skin Corr. 1B - STOT SE 3
Organes cibles
Central nervous system, Respiratory system
Risques supp
Code de la classe de stockage
4.2 - Pyrophoric and self-heating hazardous materials
Classe de danger pour l'eau (WGK)
WGK 2
Point d'éclair (°F)
31.3 °F - closed cup
Point d'éclair (°C)
-0.4 °C - closed cup
Faites votre choix parmi les versions les plus récentes :
Déjà en possession de ce produit ?
Retrouvez la documentation relative aux produits que vous avez récemment achetés dans la Bibliothèque de documents.
Les clients ont également consulté
Notre équipe de scientifiques dispose d'une expérience dans tous les secteurs de la recherche, notamment en sciences de la vie, science des matériaux, synthèse chimique, chromatographie, analyse et dans de nombreux autres domaines..
Contacter notre Service technique