S5637
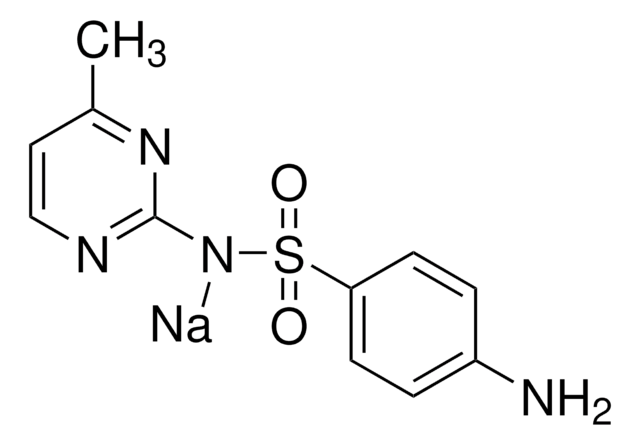
Sulfamethazine sodium salt
≥98%
Sinônimo(s):
4-Amino-N-(4,6-dimethyl-2-pyrimidinyl)benzenesulfonamide
About This Item
Produtos recomendados
Nível de qualidade
Ensaio
≥98%
Formulário
powder
condição de armazenamento
(Keep container tightly closed in a dry and well-ventilated place.)
cor
white to off-white
solubilidade
H2O: soluble 50 mg/mL
espectro de atividade do antibiótico
Gram-negative bacteria
Gram-positive bacteria
Modo de ação
DNA synthesis | interferes
enzyme | inhibits
temperatura de armazenamento
2-8°C
cadeia de caracteres SMILES
[Na].Cc1cc(C)nc(NS(=O)(=O)c2ccc(N)cc2)n1
InChI
1S/C12H14N4O2S.Na.H/c1-8-7-9(2)15-12(14-8)16-19(17,18)11-5-3-10(13)4-6-11;;/h3-7H,13H2,1-2H3,(H,14,15,16);;
chave InChI
WIVZAHIZHZEEOX-UHFFFAOYSA-N
Categorias relacionadas
Descrição geral
Aplicação
Ações bioquímicas/fisiológicas
Embalagem
Outras notas
Palavra indicadora
Warning
Frases de perigo
Declarações de precaução
Classificações de perigo
Acute Tox. 4 Oral
Código de classe de armazenamento
11 - Combustible Solids
Classe de risco de água (WGK)
WGK 3
Ponto de fulgor (°F)
Not applicable
Ponto de fulgor (°C)
Not applicable
Escolha uma das versões mais recentes:
Certificados de análise (COA)
Não está vendo a versão correta?
Se precisar de uma versão específica, você pode procurar um certificado específico pelo número do lote ou da remessa.
Já possui este produto?
Encontre a documentação dos produtos que você adquiriu recentemente na biblioteca de documentos.
Os clientes também visualizaram
Active Filters
Nossa equipe de cientistas tem experiência em todas as áreas de pesquisa, incluindo Life Sciences, ciência de materiais, síntese química, cromatografia, química analítica e muitas outras.
Entre em contato com a assistência técnica