911771
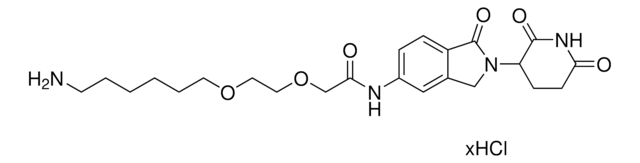
Lenalidomide-Photoswitch1-NH2 hydrochloride
≥95%
Sinônimo(s):
(E)-N-(4-Aminobutyl)-2-(4-((2-(2,6-dioxopiperidin-3-yl)-1-oxoisoindolin-4-yl)diazenyl)-2,6-dimethoxyphenoxy)acetamide hydrochloride, PHOTAC template, Photoswitchable protein degrader building block for PROTAC® research
About This Item
Produtos recomendados
ligand
lenalidomide
Nível de qualidade
Ensaio
≥95%
forma
powder or crystals
adequação da reação
reactivity: carboxyl reactive
reagent type: ligand-linker conjugate
disponibilidade
available only in USA
grupo funcional
amine
temperatura de armazenamento
2-8°C
cadeia de caracteres SMILES
NCCCCNC(COC1=C(OC)C=C(/N=N/C2=CC=CC3=C2CN(C4C(NC(CC4)=O)=O)C3=O)C=C1OC)=O.Cl
Categorias relacionadas
Aplicação
Suggested wavelengths for photoswitching:
- Switch to cis isomer: 390 nm (380-400 nm)
- Switch to trans isomer (thermally more stable isomer): >450 nm
Low-intensity light needed for photoactivation is not cytotoxic.
Product can be used with our line of photoreactors: Including Penn PhD (Z744035) & SynLED 2.0 (Z744080)
Learn more:
Outras notas
Informações legais
produto relacionado
Código de classe de armazenamento
11 - Combustible Solids
Classe de risco de água (WGK)
WGK 3
Ponto de fulgor (°F)
Not applicable
Ponto de fulgor (°C)
Not applicable
Certificados de análise (COA)
Busque Certificados de análise (COA) digitando o Número do Lote do produto. Os números de lote e remessa podem ser encontrados no rótulo de um produto após a palavra “Lot” ou “Batch”.
Já possui este produto?
Encontre a documentação dos produtos que você adquiriu recentemente na biblioteca de documentos.
Nossa equipe de cientistas tem experiência em todas as áreas de pesquisa, incluindo Life Sciences, ciência de materiais, síntese química, cromatografia, química analítica e muitas outras.
Entre em contato com a assistência técnica