146498
Trimethylzinnchlorid
Synonym(e):
Chlorotrimethylstannane
About This Item
Empfohlene Produkte
Form
crystals
mp (Schmelzpunkt)
37-39 °C (lit.)
SMILES String
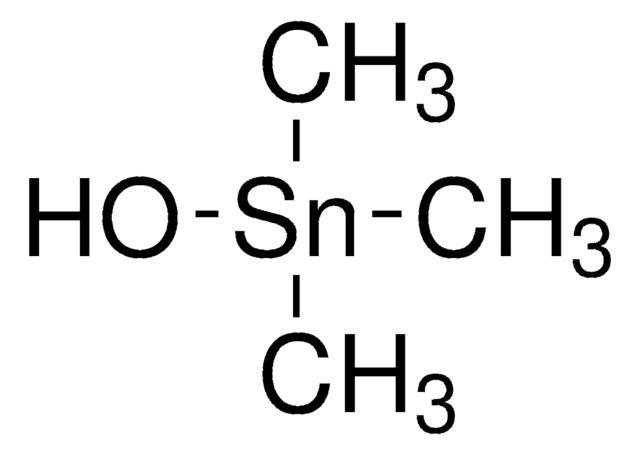
C[Sn](C)(C)Cl
InChI
1S/3CH3.ClH.Sn/h3*1H3;1H;/q;;;;+1/p-1
InChIKey
KWTSZCJMWHGPOS-UHFFFAOYSA-M
Suchen Sie nach ähnlichen Produkten? Aufrufen Leitfaden zum Produktvergleich
Verwandte Kategorien
Allgemeine Beschreibung
Anwendung
It can also be used as a reagent to prepare:
- Organotrimethyltin derivatives by reacting with organocopper compounds via transmetalation reaction.
- Acetophenone by palladium-catalyzed coupling reaction with benzoyl chloride.
- Optically active propargyl trimethylstannane by treating with chiral allenyltitanium.
- Trimethylstannyl nucleophiles, which are applicable in the formation of Sn-C bonds via SN2 reactions, SRN1 reactions, and halogen-metal exchanges.
- Carbocycles by reacting with unactivated dienes or trienes via radical-mediated carbocyclization reaction in the presence of NaBH3CN and a catalytic amount of AIBN.
Me3SnCl can also be used as a Lewis acid catalyst in asymmetric allylic alkylation reactions.
Signalwort
Danger
H-Sätze
Gefahreneinstufungen
Acute Tox. 1 Dermal - Acute Tox. 2 Inhalation - Acute Tox. 2 Oral - Aquatic Acute 1 - Aquatic Chronic 1
Lagerklassenschlüssel
6.1A - Combustible acute toxic Cat. 1 and 2 / very toxic hazardous materials
WGK
WGK 2
Flammpunkt (°F)
206.6 °F - closed cup
Flammpunkt (°C)
97 °C - closed cup
Persönliche Schutzausrüstung
Eyeshields, Faceshields, Gloves, type P3 (EN 143) respirator cartridges
Analysenzertifikate (COA)
Suchen Sie nach Analysenzertifikate (COA), indem Sie die Lot-/Chargennummer des Produkts eingeben. Lot- und Chargennummern sind auf dem Produktetikett hinter den Wörtern ‘Lot’ oder ‘Batch’ (Lot oder Charge) zu finden.
Besitzen Sie dieses Produkt bereits?
In der Dokumentenbibliothek finden Sie die Dokumentation zu den Produkten, die Sie kürzlich erworben haben.
Kunden haben sich ebenfalls angesehen
Unser Team von Wissenschaftlern verfügt über Erfahrung in allen Forschungsbereichen einschließlich Life Science, Materialwissenschaften, chemischer Synthese, Chromatographie, Analytik und vielen mehr..
Setzen Sie sich mit dem technischen Dienst in Verbindung.