Select a Size
| Pack Size | SKU | Availability | Price |
|---|---|---|---|
| 50 mg | Please contact Customer Service for Availability | $623.00 |
About This Item
ligand
A1V2PF2
Quality Level
assay
≥95%
form
crystals
reaction suitability
reactivity: carboxyl reactive, reagent type: ligand-linker conjugate
functional group
amine
storage temp.
2-8°C
SMILES string
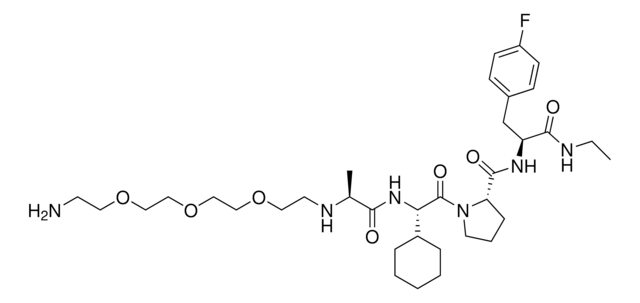
C[C@H](NCCOCCOCCOCCN)C(N[C@H](C(N1CCC[C@H]1C(N[C@H](C(NCC)=O)CC2=CC=C(C=C2)F)=O)=O)C3CCCCC3)=O
InChI
1S/C35H57FN6O7/c1-3-38-33(44)29(24-26-11-13-28(36)14-12-26)40-34(45)30-10-7-17-42(30)35(46)31(27-8-5-4-6-9-27)41-32(43)25(2)39-16-19-48-21-23-49-22-20-47-18-15-37/h11-14,25,27,29-31,39H,3-10,15-24,37H2,1-2H3,(H,38,44)(H,40,45)(H,41,43)/t25-,29-,30-,31-/m0
InChI key
BUVZIAAEQPEKLD-OVOJMXFRSA-N
Application
Building blocks in this series:
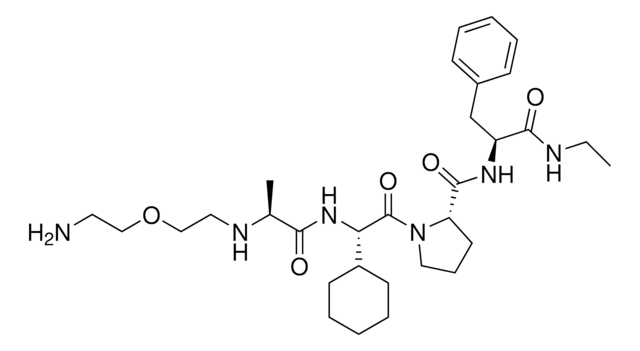
916714 A1V2PF2-NHEt
917931 A1V2PF2-NHEt-C6-NH2
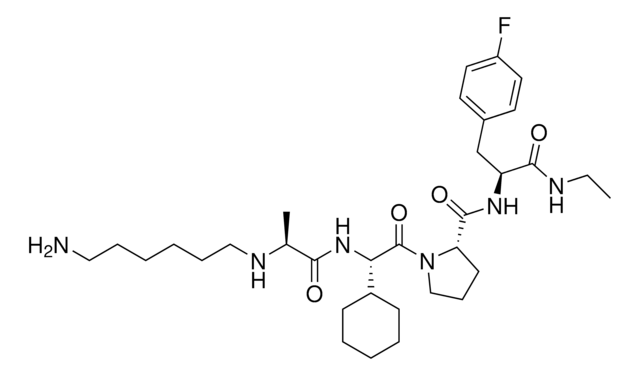
916684 A1V2PF2-NHEt-C10-NH2
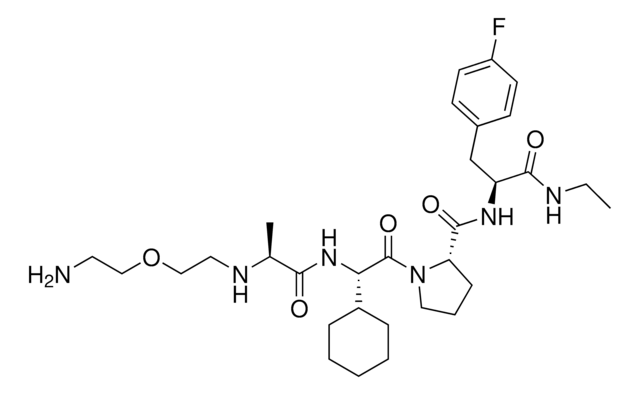
916935 A1V2PF2-NHEt-PEG1-NH2
917192 A1V2PF2-NHEt-PEG3-NH2
Technology Spotlight: Degrader Building Blocks with Inhibitor of Apoptosis Protein (IAP) In Silico-Derived Ligands
Other Notes
Legal Information
1 of 1
This Item | |||
|---|---|---|---|
| assay ≥95% | assay - | assay ≥95% | assay - |
| functional group amine | functional group amine | functional group amine | functional group amine |
| reaction suitability reactivity: carboxyl reactive, reagent type: ligand-linker conjugate | reaction suitability reactivity: carboxyl reactive, reagent type: ligand-linker conjugate | reaction suitability reactivity: carboxyl reactive, reagent type: ligand-linker conjugate | reaction suitability reactivity: carboxyl reactive, reagent type: ligand-linker conjugate |
| Quality Level 100 | Quality Level 100 | Quality Level 100 | Quality Level 100 |
| storage temp. 2-8°C | storage temp. 2-8°C | storage temp. 2-8°C | storage temp. 2-8°C |
| form crystals | form powder | form powder | form powder |
Storage Class
11 - Combustible Solids
wgk
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Articles
Plate of 80 ligands against E3 ligase IAP designed by ComInnex; allows creation of bifunctional targeted protein degraders or molecular glues.
Targeted protein degradation reduces disease-relevant proteins in cells using small molecules, hijacking endogenous proteolysis systems.
Protein Degrader Building Blocks are a collection of crosslinker-E3 ligand conjugates with a pendant functional group for covalent linkage to a target ligand.
Global Trade Item Number
| SKU | GTIN |
|---|---|
| 917192-50MG | 04061842653850 |