推薦產品
等級
pharmaceutical primary standard
API 家族
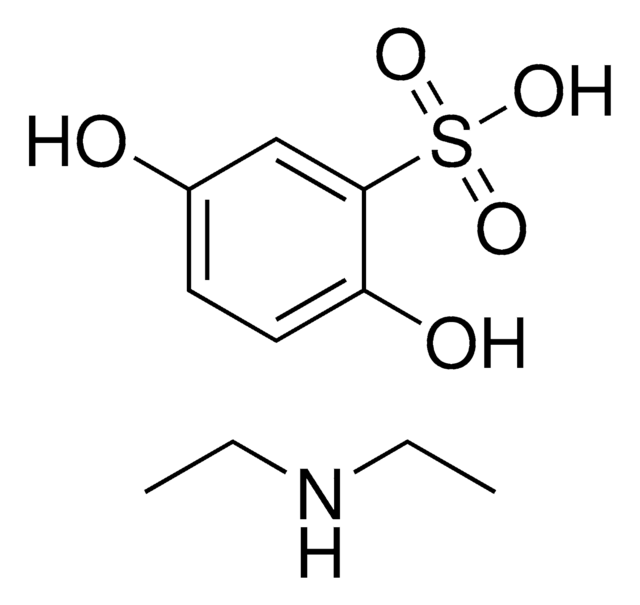
etamsylate
製造商/商標名
EDQM
應用
pharmaceutical (small molecule)
格式
neat
儲存溫度
2-8°C
InChI
1S/C6H6O5S.C4H11N/c7-4-1-2-5(8)6(3-4)12(9,10)11;1-3-5-4-2/h1-3,7-8H,(H,9,10,11);5H,3-4H2,1-2H3
InChI 密鑰
HBGOLJKPSFNJSD-UHFFFAOYSA-N
尋找類似的產品? 前往 產品比較指南
一般說明
This product is provided as delivered and specified by the issuing Pharmacopoeia. All information provided in support of this product, including SDS and any product information leaflets have been developed and issued under the Authority of the issuing Pharmacopoeia.For further information and support please go to the website of the issuing Pharmacopoeia.
應用
Etamsylate EP Reference standard, intended for use in laboratory tests only as specifically prescribed in the European Pharmacopoeia.
包裝
The product is delivered as supplied by the issuing Pharmacopoeia. For the current unit quantity, please visit the EDQM reference substance catalogue.
其他說明
Sales restrictions may apply.
儲存類別代碼
11 - Combustible Solids
水污染物質分類(WGK)
WGK 3
閃點(°F)
Not applicable
閃點(°C)
Not applicable
Ondrej Wiewiorka et al.
Scandinavian journal of clinical and laboratory investigation, 73(5), 449-451 (2013-05-22)
With discrepancies encountered as early as the verification of enzymatic method for quantification of serum creatinine, our research pointed to a later confirmed interference caused by a compound called ethamsylate present in the commonly used antihemorrhagic drug Dicynone. We measured
Alaa El-Gindy et al.
Journal of AOAC International, 96(4), 730-736 (2013-09-05)
A simple, selective, sensitive, accurate, and precise method was developed for determination of ethamsylate (ET) in human urine and in ET tablets using RP-HPLC. The method uses a C18 (5 pm particle size) column at ambient temperature with the mobile
Takehito Naruoka et al.
Hinyokika kiyo. Acta urologica Japonica, 54(5), 341-344 (2008-06-13)
We examined the effectiveness of supplemental administration of Eviprostat in patients with benign prostatic hyperplasia (BPH) whose lower urinary tract symptoms (LUTS) caused by BPH were not adequately relieved by an alpha1-adrenoceptor blocker. Twenty-nine patients with insufficient improvement in the
Rod Hunt et al.
The Cochrane database of systematic reviews, (1)(1), CD004343-CD004343 (2010-01-22)
Ethamsylate decreases blood loss in certain clinical situations such as menorrhagia and following some surgical procedures. This potential to reduce bleeding has led to the hypothesis that it may have a role to play in reducing intraventricular haemorrhage in preterm
Hiroyuki Tsunemori et al.
Urology, 77(6), 1507-1507 (2011-06-01)
To examine the effect of the phytotherapeutic agent Eviprostat on the stromal-to-epithelial (S/E) ratio, level of macrophage infiltration, expression of the macrophage inhibitory cytokine-1 (Mic1) gene, and tumor necrosis factor-alpha (TNF-α) and interleukin-8 (IL-8) concentrations in prostate tissues in a
我們的科學家團隊在所有研究領域都有豐富的經驗,包括生命科學、材料科學、化學合成、色譜、分析等.
聯絡技術服務