推薦產品
化驗
97%
mp
66-70 °C (lit.)
官能基
aldehyde
SMILES 字串
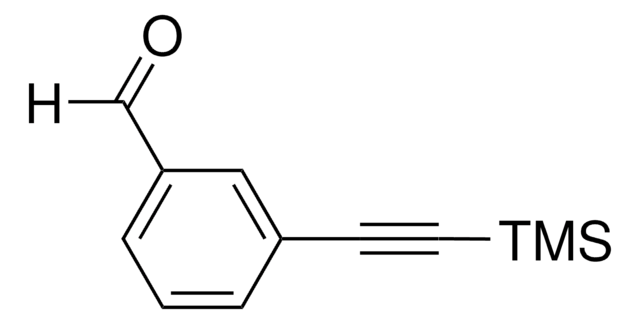
C[Si](C)(C)C#Cc1ccc(C=O)cc1
InChI
1S/C12H14OSi/c1-14(2,3)9-8-11-4-6-12(10-13)7-5-11/h4-7,10H,1-3H3
InChI 密鑰
UZQDUXAJFTWMDT-UHFFFAOYSA-N
一般說明
4-[(Trimethylsilyl)ethynyl]benzaldehyde can be synthesized by reacting 4-bromobenzaldehyde and triphenylphosphine in anhydrous triethylamine with ethynyltrimethylsilane and then with palladium(II) acetate under argon.[1] It can also be prepared by Sonogashira coupling reaction between 4-bromobenzaldehyde and (trimethylsilyl) ethynyl.[2]
儲存類別代碼
11 - Combustible Solids
水污染物質分類(WGK)
WGK 3
閃點(°F)
Not applicable
閃點(°C)
Not applicable
個人防護裝備
Eyeshields, Gloves, type N95 (US)
從最近期的版本中選擇一個:
分析證明 (COA)
Lot/Batch Number
客戶也查看了
Facile synthesis of ethynylated benzoic acid derivatives and aromatic compounds via ethynyltrimethylsilane.
Austin WB, et al.
The Journal of Organic Chemistry, 46(11), 2280-2286 (1981)
On the potential of porphyrin-spiked triarylamine stars for bulk heterojunction solar cells.
Kengthanomma T, et al.
Journal of Material Chemistry A, 1(35), 10524-10531 (2013)
Linear porphyrin dimers with fluorenyl arms linked by an ethynyl bridge.
Merhi A, et al.
Tetrahedron, 69(34), 7112-7124 (2013)
Synthesis of covalently bonded nanostructure from two porphyrin molecular wires leading to a molecular tube.
Ishida T, et al.
Tetrahedron Letters, 47(30), 5265-5268 (2006)
Cascade Reactions of Me3Si-Substituted Imidazolidine-1, 3-Diols with PbO2, Including Oxidation of the Corresponding Diol and Subsequent Elimination of the Trimethylsilyl Fragment.
Tretyakov E, et al.
European Journal of Organic Chemistry, 2007(22), 3639-3647 (2007)
Active Filters
我們的科學家團隊在所有研究領域都有豐富的經驗,包括生命科學、材料科學、化學合成、色譜、分析等.
聯絡技術服務

![2-[(三甲基硅基)乙炔基]苯甲醛 97%](/deepweb/assets/sigmaaldrich/product/structures/977/954/5bc6c647-e9d2-4bf6-bf94-6ac20e3f476e/640/5bc6c647-e9d2-4bf6-bf94-6ac20e3f476e.png)








