推薦產品
同位素純度
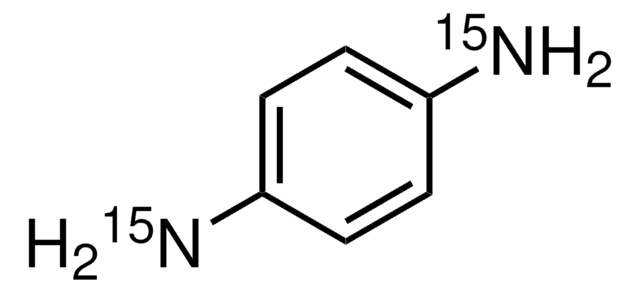
98 atom % 15N
品質等級
化驗
99% (CP)
折射率
n20/D 1.586 (lit.)
bp
184 °C (lit.)
mp
−6 °C (lit.)
密度
1.033 g/mL at 25 °C
質量偏移
M+1
儲存溫度
2-8°C
SMILES 字串
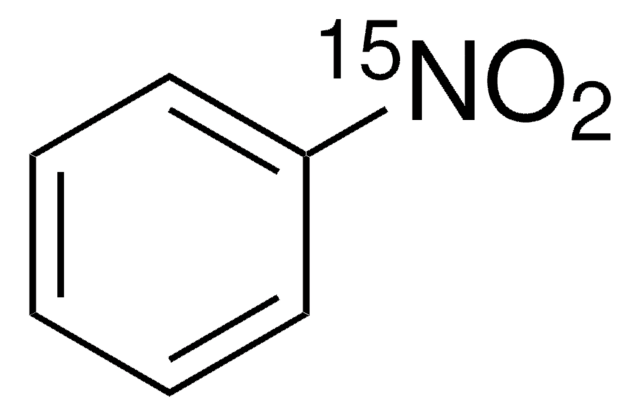
[15NH2]c1ccccc1
InChI
1S/C6H7N/c7-6-4-2-1-3-5-6/h1-5H,7H2/i7+1
InChI 密鑰
PAYRUJLWNCNPSJ-CDYZYAPPSA-N
包裝
This product may be available from bulk stock and can be packaged on demand. For information on pricing, availability and packaging, please contact Stable Isotopes Customer Service.
訊號詞
Danger
危險分類
Acute Tox. 3 Dermal - Acute Tox. 3 Inhalation - Acute Tox. 3 Oral - Aquatic Acute 1 - Aquatic Chronic 1 - Carc. 2 - Eye Dam. 1 - Muta. 2 - Resp. Sens. 1
儲存類別代碼
6.1A - Combustible acute toxic Cat. 1 and 2 / very toxic hazardous materials
水污染物質分類(WGK)
WGK 3
閃點(°F)
158.0 °F - closed cup
閃點(°C)
70 °C - closed cup
客戶也查看了
Conghui Tang et al.
Journal of the American Chemical Society, 134(46), 18924-18927 (2012-11-08)
A novel and efficient copper-catalyzed azidation reaction of anilines via C-H activation has been developed. This method, in which the primary amine acts as a directing group by coordinating to the metal center, provides ortho azidation products regioselectively under mild
Timo Stahl et al.
Journal of the American Chemical Society, 135(4), 1248-1251 (2013-01-15)
Heterolytic splitting of the Si-H bond mediated by a Ru-S bond forms a sulfur-stabilized silicon cation that is sufficiently electrophilic to abstract fluoride from CF(3) groups attached to selected anilines. The ability of the Ru-H complex, generated in the cooperative
Dongdong Liang et al.
Chemical communications (Cambridge, England), 49(2), 173-175 (2012-11-22)
An efficient synthesis of free (NH)-phenanthridinones through Pd-catalyzed C(sp(2))-H aminocarbonylation of unprotected o-arylanilines under an atmospheric pressure of CO has been developed. Some ortho heteroarene substituted anilines as well as N-alkyl protected o-arylanilines are also suitable substrates for this C-H
Sung Jin Bae et al.
European journal of medicinal chemistry, 57, 383-390 (2012-11-15)
We attempted to design and synthesize (E)-N-substituted benzylidene-hydroxy or methoxy-aniline derivatives and to evaluate their inhibitory effect on tyrosinase activity and anti-melanogenesis activity in murine B16F10 melanoma cells. Derivatives with a 4-methoxy- or 4-hydroxy-anilino group exerted more potent inhibition against
Palladium-catalyzed insertion of α-diazocarbonyl compounds for the synthesis of cyclic amino esters.
Ping-Xin Zhou et al.
Chemical communications (Cambridge, England), 49(6), 561-563 (2012-12-05)
Two different cyclic amino esters are synthesized by palladium-catalyzed cross-coupling reaction of diazoesters with N-substituted-2-iodoanilines. Aryldiazoacetates lead to cyclic α-amino esters with an α-quaternary carbon centre in the presence of CO. Additionally, arylvinyldiazoacetates afford cyclic α,β-unsaturated γ-amino esters.
Active Filters
我們的科學家團隊在所有研究領域都有豐富的經驗,包括生命科學、材料科學、化學合成、色譜、分析等.
聯絡技術服務