5.05766
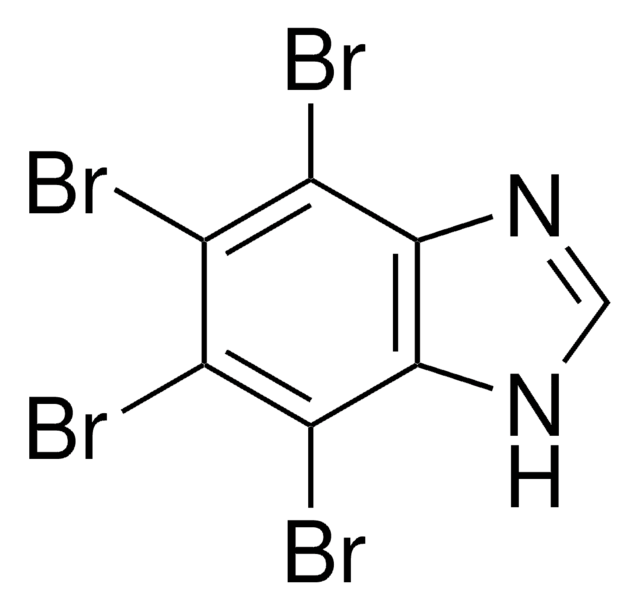
Casein Kinase II Inhibitor XII, TBBz
Synonim(y):
Casein Kinase II Inhibitor XII, TBBz, 4,5,6,7-Tetrabromobenzimidazole, CYP24A1 inhibitor, TBBz, CK2 Inhibitor, TBBz
About This Item
Polecane produkty
Próba
≥99% (HPLC)
Poziom jakości
Formularz
powder
siła działania
70 nM Ki
producent / nazwa handlowa
Calbiochem®
warunki przechowywania
OK to freeze
protect from light
kolor
off-white
rozpuszczalność
DMSO: 100 mg/mL
temp. przechowywania
2-8°C
InChI
1S/C7H2Br4N2/c8-2-3(9)5(11)7-6(4(2)10)12-1-13-7/h1H,(H,12,13)
Klucz InChI
LOEIRDBRYBHAJB-UHFFFAOYSA-N
Opis ogólny
Działania biochem./fizjol.
CK2
Opakowanie
Ostrzeżenie
Rekonstytucja
Inne uwagi
Zien, P. et al. 2003. Biochem. Biophys. Res. Comm.306, 129.
Zien, P. et al. 2003. Biochem. Biophys. Res. Comm.312, 623.
Zien, P. et al. 2005. Biochim. Biophy. Acta1754, 271.
Luo, W. et al. 2013. Cancer Res.73, 2289.
Informacje prawne
Kod klasy składowania
11 - Combustible Solids
Klasa zagrożenia wodnego (WGK)
WGK 3
Temperatura zapłonu (°F)
Not applicable
Temperatura zapłonu (°C)
Not applicable
Certyfikaty analizy (CoA)
Poszukaj Certyfikaty analizy (CoA), wpisując numer partii/serii produktów. Numery serii i partii można znaleźć na etykiecie produktu po słowach „seria” lub „partia”.
Masz już ten produkt?
Dokumenty związane z niedawno zakupionymi produktami zostały zamieszczone w Bibliotece dokumentów.
Nasz zespół naukowców ma doświadczenie we wszystkich obszarach badań, w tym w naukach przyrodniczych, materiałoznawstwie, syntezie chemicznej, chromatografii, analityce i wielu innych dziedzinach.
Skontaktuj się z zespołem ds. pomocy technicznej