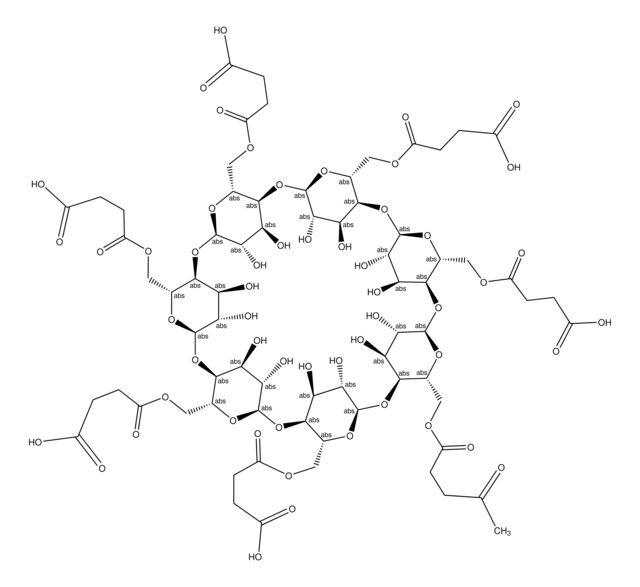
The molecular weight for the β-Cyclodextrin moiety is 1135 g/mole and the contribution from the succinic acid portion is 100.1 g/mole. This mass is used for succinic acid as water is lost during the condensation reaction when cyclodextrin and succinic acid form the conjugate. Thus, the average molecular formula and average formula weight can be expressed as the following. C42H70-nO35 • (C4H5O3)n or 1135.0 + n •100.1 The average degree of substitution for this product is ~3.5 for this product making the molecular weight approximately ~1485 g/mole for this product. The degree of substitution can vary from 2.5 - 5 for this product and the batch specific certificate of analysis should be reviewed to gather the degree of substitution (n).
Scegli un formato
| A voi/SKU | Disponibilità | Prezzo |
|---|---|---|
500 mg | Per informazioni sulla disponibilità, contatta il Servizio Clienti. | 230,00 € |
5 g | Per informazioni sulla disponibilità, contatta il Servizio Clienti. | 1.480,00 € |
Informazioni su questo articolo
230,00 €
form
powder
Quality Segment
impurities
~5% water
color
white
mp
225 °C ((437 °F ) - Decomposes on heating)
storage temp.
−20°C
SMILES string
CC(=O)CCC(=O)OC[C@H]1O[C@@H]2O[C@H]3[C@@H](O)[C@H](O)[C@H](O[C@@H]3COC(=O)CCC(O)=O)O[C@H]4[C@@H](O)[C@H](O)[C@H](O[C@@H]4COC(=O)CCC(O)=O)O[C@H]5[C@@H](O)[C@H](O)[C@H](O[C@@H]5COC(=O)CCC(O)=O)O[C@H]6[C@@H](O)[C@H](O)[C@H](O[C@@H]6COC(=O)CCC(O)=O)O[C@H]7[C@@H](O)[C@H](O)[C@H](O[C@@H]7COC(=O)CCC(O)=O)O[C@H]8[C@@H](O)[C@H](O)[C@H](O[C@@H]8COC(=O)CCC(O)=O)O[C@H]1[C@@H](O)[C@@H]2O
InChI
1S/C71H100O55/c1-23(72)2-9-37(85)106-16-24-58-44(92)51(99)65(113-24)121-59-25(17-107-38(86)10-3-31(73)74)115-67(53(101)46(59)94)123-61-27(19-109-40(88)12-5-33(77)78)117-69(55(103)48(61)96)125-63-29(21-111-42(90)14-7-35(81)82)119-71(57(105)50(63)98)126-64-30(22-112-43(91)15-8-36(83)84)118-70(56(104)49(64)97)124-62-28(20-110-41(89)13-6-34(79)80)116-68(54(102)47(62)95)122-60-26(18-108-39(87)11-4-32(75)76)114-66(120-58)52(100)45(60)93/h24-30,44-71,92-105H,2-22H2,1H3,(H,73,74)(H,75,76)(H,77,78)(H,79,80)(H,81,82)(H,83,84)/t24-,25-,26-,27-,28-,29-,30-,44+,45+,46+,47+,48+,49+,50+,51+,52+,53+,54+,55+,56+,57+,58-,59-,60-,61-,62-,63-,64-,65-,66-,67-,68-,69-,70-,71-/m1/s1
InChI key
DIRLEDPEXJLCIL-JCWBWLHSSA-N
General description
Application
Succinyl-β-cyclodextrin and carboxymethyl-β-cyclodextrin are used as chiral selective agents in capillary electrophoresis for the separation of di- and tri-peptide enantiomers and catechin enantiomers. Succinyl-β-cyclodextrin is used to optimize analysis of PNA-DNA duplexes with diethylthiadicarbocyanine dye.
Other Notes
1 of 1
Questo articolo | |||
|---|---|---|---|
| Quality Level 100 | Quality Level 200 | Quality Level 100 | Quality Level 200 |
| form powder | form powder | form solid | form powder |
| storage temp. −20°C | storage temp. 2-8°C | storage temp. - | storage temp. - |
| impurities ~5% water | impurities - | impurities 10% water | impurities - |
| color white | color - | color - | color - |
| mp 225 °C ((437 °F ) - Decomposes on heating) | mp 170-178 °C (lit.) | mp ~245 °C (dec.) | mp 290-300 °C (dec.) (lit.) |
Classe di stoccaggio
11 - Combustible Solids
wgk
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
Eyeshields, Gloves, type N95 (US)
Scegli una delle versioni più recenti:
Possiedi già questo prodotto?
I documenti relativi ai prodotti acquistati recentemente sono disponibili nell’Archivio dei documenti.
-
What is the molecular weight of this product?
1 risposta-
Utile?
-