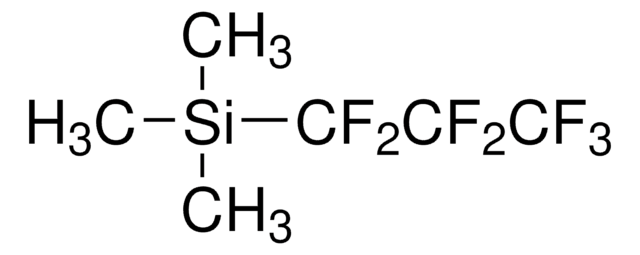900015
Trimethylpentafluoroethylsilane
97%
Sinonimo/i:
(Pentafluoroethyl)trimethylsilane
About This Item
Prodotti consigliati
Saggio
97%
Forma fisica
liquid
Indice di rifrazione
n/D 1.325
Densità
1.095 g/mL
Stringa SMILE
C[Si](C)(C)C(F)(F)C(F)(F)F
InChI
1S/C5H9F5Si/c1-11(2,3)5(9,10)4(6,7)8/h1-3H3
MTPVUVINMAGMJL-UHFFFAOYSA-N
Descrizione generale
Applicazioni
- 5-bromo-2-(perfluoroethyl)quinoline
- 8-methoxy-2-(perfluoroethyl)quinoline
- 1-(perfluoroethyl)isoquinoline
- 8-(tert-butoxy)-5,7-dichloro-2-(perfluoroethyl)quinolone
Avvertenze
Danger
Indicazioni di pericolo
Consigli di prudenza
Classi di pericolo
Eye Irrit. 2 - Flam. Liq. 2 - Skin Irrit. 2 - STOT SE 3
Organi bersaglio
Respiratory system
Codice della classe di stoccaggio
3 - Flammable liquids
Classe di pericolosità dell'acqua (WGK)
WGK 3
Punto d’infiammabilità (°F)
1.4 °F - closed cup - (calculated)
Punto d’infiammabilità (°C)
-17 °C - closed cup - (calculated)
Certificati d'analisi (COA)
Cerca il Certificati d'analisi (COA) digitando il numero di lotto/batch corrispondente. I numeri di lotto o di batch sono stampati sull'etichetta dei prodotti dopo la parola ‘Lotto’ o ‘Batch’.
Possiedi già questo prodotto?
I documenti relativi ai prodotti acquistati recentemente sono disponibili nell’Archivio dei documenti.
Contenuto correlato
The major research interests of Prof. Jinbo Hu's lab include the development of new fluorination reagents and reactions, especially the difluoromethylation, difluoromethylenation, and monofluoromethylation methods.
Il team dei nostri ricercatori vanta grande esperienza in tutte le aree della ricerca quali Life Science, scienza dei materiali, sintesi chimica, cromatografia, discipline analitiche, ecc..
Contatta l'Assistenza Tecnica.