推荐产品
方案
≥98%
mp
245-247 °C (lit.)
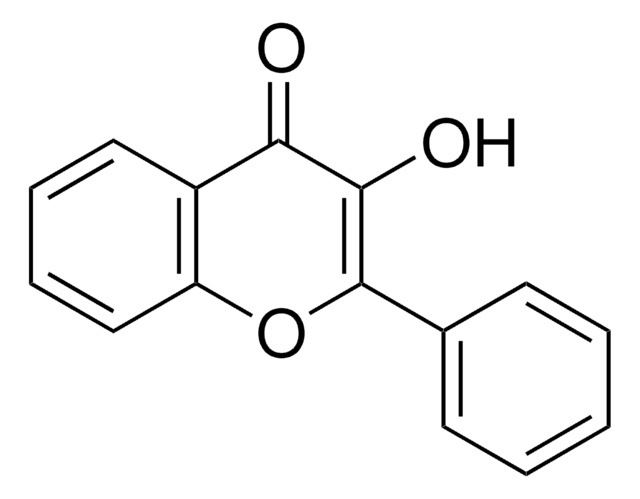
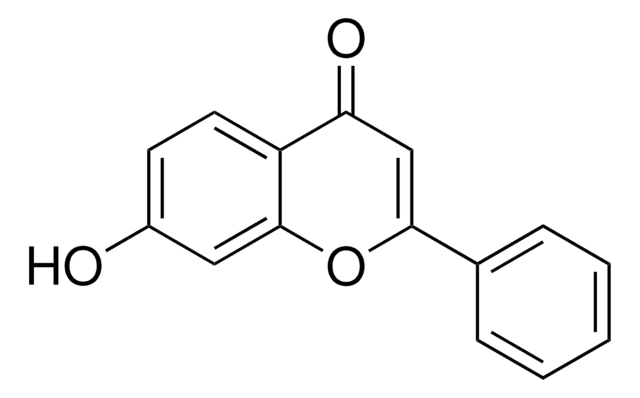
SMILES字符串
Oc1ccc2C(=O)C=C(Oc2c1)c3ccccc3
InChI
1S/C15H10O3/c16-11-6-7-12-13(17)9-14(18-15(12)8-11)10-4-2-1-3-5-10/h1-9,16H
InChI key
MQGPSCMMNJKMHQ-UHFFFAOYSA-N
基因信息
mouse ... Hexa(15211)
rat ... Ar(24208) , Gabra2(29706)
正在寻找类似产品? 访问 产品对比指南
应用
作为如下过程的反应物:
- 在 O-糖基化反应中作为具有生物学价值的受体
- 参与合成完全磷酸化的黄酮类化合物,用作胰腺胆固醇酯酶抑制剂
- 用于碳酸二甲酯的 O-甲基化
- 通过多亚甲基链连接,合成 α1-肾上腺素受体拮抗剂
- 参与 Baylis-Hillman 反应
- 参与相转移催化的糖基化反应,合成糖基化的类黄酮
警示用语:
Warning
危险声明
危险分类
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
靶器官
Respiratory system
储存分类代码
11 - Combustible Solids
WGK
WGK 3
个人防护装备
dust mask type N95 (US), Eyeshields, Gloves
其他客户在看
Osamu Kagami et al.
Journal of bioscience and bioengineering, 106(2), 121-127 (2008-09-23)
A central part (amino-acid position 268-397 of 458 amino-acid residues) of the biphenyl dioxygenase large (alpha) subunit, BphA1, from Pseudomonas pseudoalcaligenes strain KF707 was exchanged with the corresponding part of BphA1 from another biphenyl-degrading bacterium, Pseudomonas putida strain KF715, to
Jianbo Xiao et al.
Molecular nutrition & food research, 54 Suppl 2, S253-S260 (2010-03-23)
Four flavones (flavone, 7-hydroxyflavone, chrysin, and baicalein) sharing the same B- and C-ring structure but a different numbers of hydroxyl groups on the A-ring were studied for their affinities for BSA and HSA. The hydroxylation on ring A of flavones
Nga Ta et al.
The Journal of steroid biochemistry and molecular biology, 107(1-2), 127-129 (2007-07-13)
Previous studies have shown chrysin, 7-hydroxyflavone and 7,4'-dihydroxyflavone to be the most potent flavonoid inhibitors of aromatase. However, very poor oral bioavailability is a major limitation for the successful use of dietary flavonoids as chemopreventive agents. We have recently shown
T K Vinh et al.
Journal of enzyme inhibition, 16(5), 417-424 (2002-03-28)
A series of 4-aryl substituted 7-hydroxy-flavones were prepared using the three-step Baker-Venkataraman synthesis in good overall yields. The flavones were all evaluated in vitro for inhibitory activity against aromatase (P450AROM, CYP19), using human placental microsomes, and for inhibitory activity against
R Le Lain et al.
Journal of enzyme inhibition, 16(1), 35-45 (2001-08-11)
In a screening programme for inhibitors of human testis 17beta-hydroxysteroid dehydrogenase (17beta-HSD type 3), as potential agents for the treatment of hormone-dependent prostatic cancer, we have used crude human testis microsomal 17beta-hydroxysteroid dehydrogenase as a convenient source of the enzyme.
我们的科学家团队拥有各种研究领域经验,包括生命科学、材料科学、化学合成、色谱、分析及许多其他领域.
联系客户支持