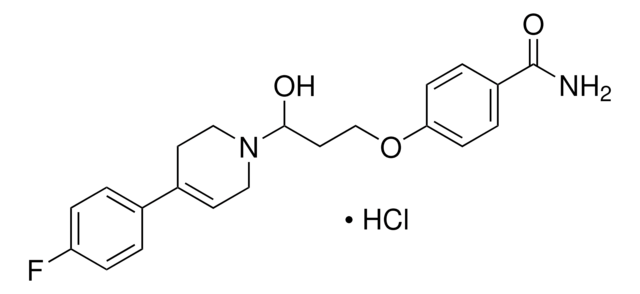
I119
Indatraline hydrochloride
solid
Synonyma:
(±)-trans-3-(3,4-Dichlorophenyl)-N-methyl-1-indanamine hydrochloride, Lu 19-005
About This Item
Doporučené produkty
form
solid
color
white
solubility
H2O: 2 mg/mL
storage temp.
2-8°C
SMILES string
Cl.CN[C@@H]1C[C@@H](c2ccc(Cl)c(Cl)c2)c3ccccc13
InChI
1S/C16H15Cl2N.ClH/c1-19-16-9-13(11-4-2-3-5-12(11)16)10-6-7-14(17)15(18)8-10;/h2-8,13,16,19H,9H2,1H3;1H/t13-,16+;/m0./s1
InChI key
QICQDZXGZOVTEF-MELYUZJYSA-N
Gene Information
human ... DRD1(1812) , DRD2(1813) , DRD3(1814) , DRD4(1815) , DRD5(1816) , HTR1A(3350) , HTR1B(3351) , HTR1D(3352) , HTR1E(3354) , HTR1F(3355) , HTR2A(3356) , HTR2B(3357) , HTR2C(3358) , HTR3A(3359) , HTR3B(9177) , HTR3C(170572) , HTR3D(200909) , HTR3E(285242) , HTR4(3360) , HTR5A(3361) , HTR5B(645694) , HTR6(3362) , HTR7(3363)
Application
- as a competitive inhibitor of 3H-dopamine ([3H]DA) to study its effects on trans-activator of transcription (Tat) protein on cocaine-induced inhibition of uptake of [3H]DA
- as a dopamine transport blocker to study its effects on trace amine-associated receptor 1 (TAAR1)-transfected mice cells
- as a nonselective monoamine transport inhibitor to study its anti-angiogenic activities in glioblastoma multiforme (GBM)
Biochem/physiol Actions
Features and Benefits
Legal Information
signalword
Warning
hcodes
pcodes
Hazard Classifications
Aquatic Acute 1 - Aquatic Chronic 1
Storage Class
11 - Combustible Solids
wgk_germany
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
Gloves
Osvědčení o analýze (COA)
Vyhledejte osvědčení Osvědčení o analýze (COA) zadáním čísla šarže/dávky těchto produktů. Čísla šarže a dávky lze nalézt na štítku produktu za slovy „Lot“ nebo „Batch“.
Již tento produkt vlastníte?
Dokumenty související s produkty, které jste v minulosti zakoupili, byly za účelem usnadnění shromážděny ve vaší Knihovně dokumentů.
Náš tým vědeckých pracovníků má zkušenosti ve všech oblastech výzkumu, včetně přírodních věd, materiálových věd, chemické syntézy, chromatografie, analytiky a mnoha dalších..
Obraťte se na technický servis.