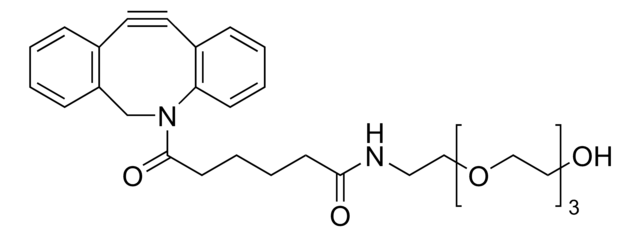
761524
Dibenzocyclooctyne-N-hydroxysuccinimidyl ester
for Copper-free Click Chemistry
Sinonimo/i:
DBCO-NHS ester, DBCO-SE
About This Item
Prodotti consigliati
Forma fisica
solid
Impiego in reazioni chimiche
reaction type: click chemistry
reagent type: cross-linking reagent
Punto di fusione
149-157 (decomposition)
Gruppo funzionale
NHS ester
Temperatura di conservazione
−20°C
Stringa SMILE
O=C(CCC(ON(C(CC1)=O)C1=O)=O)N2CC3=C(C=CC=C3)C#CC4=C2C=CC=C4
InChI
1S/C23H18N2O5/c26-20(13-14-23(29)30-25-21(27)11-12-22(25)28)24-15-18-7-2-1-5-16(18)9-10-17-6-3-4-8-19(17)24/h1-8H,11-15H2
XCEBOJWFQSQZKR-UHFFFAOYSA-N
Descrizione generale
Applicazioni
- In the modification and labeling of biomolecules such as antibodies and streptavidin by introducing DBCO groups onto their surfaces
- As a cross linker to conjugate peptide antigens onto the surface of poly(lactic-co-glycolic acid) (PLGA) nanoparticles
Codice della classe di stoccaggio
11 - Combustible Solids
Classe di pericolosità dell'acqua (WGK)
WGK 3
Punto d’infiammabilità (°F)
Not applicable
Punto d’infiammabilità (°C)
Not applicable
Certificati d'analisi (COA)
Cerca il Certificati d'analisi (COA) digitando il numero di lotto/batch corrispondente. I numeri di lotto o di batch sono stampati sull'etichetta dei prodotti dopo la parola ‘Lotto’ o ‘Batch’.
Possiedi già questo prodotto?
I documenti relativi ai prodotti acquistati recentemente sono disponibili nell’Archivio dei documenti.
I clienti hanno visto anche
Articoli
Copper-free click chemistry is an alternative approach to click chemistry that proceeds at a lower activation barrier and is free of cytotoxic transition metal catalysts.
Drug discovery process by utilizing chemistry reaction of Cu(I)-catalyzed Huisgen 1,3-dipolar cycloaddition of terminal alkynes with organoazides to yield 1,4-disubstituted 1,2,3-triazoles.
Il team dei nostri ricercatori vanta grande esperienza in tutte le aree della ricerca quali Life Science, scienza dei materiali, sintesi chimica, cromatografia, discipline analitiche, ecc..
Contatta l'Assistenza Tecnica.



![(1R,8S,9s)-Bicyclo[6.1.0]non-4-yn-9-ylmethyl N-succinimidyl carbonate for Copper-free Click Chemistry](/deepweb/assets/sigmaaldrich/product/structures/969/022/d6776082-2f7a-47c7-bcd4-3830dac0fb7d/640/d6776082-2f7a-47c7-bcd4-3830dac0fb7d.png)

![N-[(1R,8S,9s)-Bicyclo[6.1.0]non-4-yn-9-ylmethyloxycarbonyl]-1,8-diamino-3,6-dioxaoctane for Copper-free Click Chemistry](/deepweb/assets/sigmaaldrich/product/structures/294/853/c5e47d84-5aee-4797-aa24-604f291171cc/640/c5e47d84-5aee-4797-aa24-604f291171cc.png)



![(1R,8S,9s)-Bicyclo[6.1.0]non-4-yn-9-ylmethanol for Copper-free Click Chemistry](/deepweb/assets/sigmaaldrich/product/structures/171/632/0556139a-2db5-4678-a6ec-a26a693fd574/640/0556139a-2db5-4678-a6ec-a26a693fd574.png)