Wichtige Dokumente
M6760
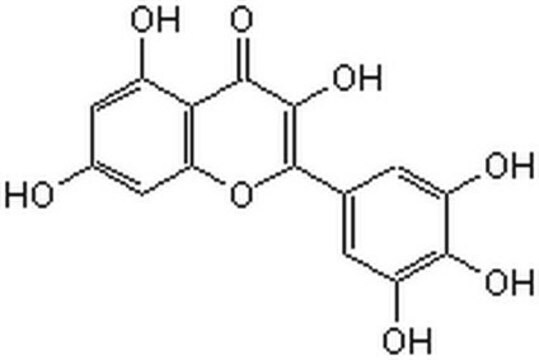
Myricetin
≥96.0%, crystalline
Synonym(e):
3,3′,4′,5,5′,7-Hexahydroxy-flavon, Cannabiscetin, Myricetol
About This Item
Empfohlene Produkte
Qualitätsniveau
Assay
≥96.0%
Form
crystalline
mp (Schmelzpunkt)
>300 °C (lit.)
Löslichkeit
absolute ethanol: 10 mg/mL, clear to slightly hazy, yellow to very deep greenish-yellow
SMILES String
Oc1cc(O)c2C(=O)C(O)=C(Oc2c1)c3cc(O)c(O)c(O)c3
InChI
1S/C15H10O8/c16-6-3-7(17)11-10(4-6)23-15(14(22)13(11)21)5-1-8(18)12(20)9(19)2-5/h1-4,16-20,22H
InChIKey
IKMDFBPHZNJCSN-UHFFFAOYSA-N
Angaben zum Gen
human ... CYP1A2(1544)
mouse ... Hexa(15211)
rat ... Il4(287287) , Tnf(24835)
Suchen Sie nach ähnlichen Produkten? Aufrufen Leitfaden zum Produktvergleich
Anwendung
- to investigate its effect on end product (AGE)- bovine serum albumin mediated phosphorylation of mitogen-activated protein kinase(ERK1) [1]
- as a standard for the quantification of phenolics from noni plant extracts using high performance liquid chromatography(HPLC)[2]
- as a standard for characterization of phenolic compounds from Hibiscus sabdariffa using ultra-high performance liquid chromatography(UHPLC)[3]
Biochem./physiol. Wirkung
Lagerklassenschlüssel
11 - Combustible Solids
WGK
WGK 3
Flammpunkt (°F)
Not applicable
Flammpunkt (°C)
Not applicable
Persönliche Schutzausrüstung
Eyeshields, Gloves, type N95 (US)
Hier finden Sie alle aktuellen Versionen:
Besitzen Sie dieses Produkt bereits?
In der Dokumentenbibliothek finden Sie die Dokumentation zu den Produkten, die Sie kürzlich erworben haben.
Kunden haben sich ebenfalls angesehen
Artikel
Antioxidants protect biological systems from oxidative damage produced by oxygen-containing free radicals and from redoxactive transition metal ions such as iron, copper, and cadmium.
Protokolle
Protocol for HPLC Analysis of Flavonoids on Ascentis® RP-Amide
Active Filters
Unser Team von Wissenschaftlern verfügt über Erfahrung in allen Forschungsbereichen einschließlich Life Science, Materialwissenschaften, chemischer Synthese, Chromatographie, Analytik und vielen mehr..
Setzen Sie sich mit dem technischen Dienst in Verbindung.