Products may be shipped at a different temperature than the recommended long-term storage temperature. If the product quality is sensitive to short-term exposure to conditions other than the recommended long-term storage, it will be shipped on wet or dry-ice. If the product quality is NOT affected by short-term exposure to conditions other than the recommended long-term storage, it will be shipped at ambient temperature. As shipping routes are configured for minimum transit times, shipping at ambient temperature helps control shipping costs for our customers. For more information, please refer to the Storage and Transport Conditions document: https://www.sigmaaldrich.com/deepweb/assets/sigmaaldrich/marketing/global/documents/316/622/storage-transport-conditions-mk.pdf
Wichtige Dokumente
PHR1431
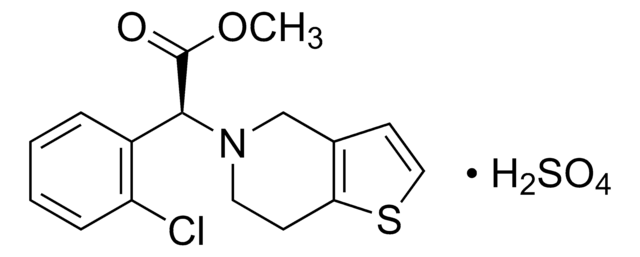
Clopidogrel-Bisulfat
Pharmaceutical Secondary Standard; Certified Reference Material
Synonym(e):
(S)-(2-Chlorphenyl)(6,7-dihydro-4H-thieno[3,2-c]pyridin-5-yl)-essigsäure-methylester -hydrogensulfat
Größe auswählen
About This Item
Empfohlene Produkte
Qualität
certified reference material
pharmaceutical secondary standard
Qualitätsniveau
Agentur
traceable to Ph. Eur. Y0001333
traceable to USP 1140430
API-Familie
clopidogrel
Analysenzertifikat (CofA)
current certificate can be downloaded
Verpackung
pkg of 1 g
Methode(n)
HPLC: suitable
gas chromatography (GC): suitable
Anwendung(en)
pharmaceutical (small molecule)
Format
neat
Lagertemp.
2-8°C
SMILES String
OS(O)(=O)=O.COC(=O)[C@@H](N1CCc2sccc2C1)c3ccccc3Cl
InChI
1S/C16H16ClNO2S.H2O4S/c1-20-16(19)15(12-4-2-3-5-13(12)17)18-8-6-14-11(10-18)7-9-21-14;1-5(2,3)4/h2-5,7,9,15H,6,8,10H2,1H3;(H2,1,2,3,4)/t15-;/m0./s1
InChIKey
FDEODCTUSIWGLK-RSAXXLAASA-N
Angaben zum Gen
human ... P2RY12(64805)
Suchen Sie nach ähnlichen Produkten? Aufrufen Leitfaden zum Produktvergleich
Allgemeine Beschreibung
Clopidogrel Bisulfate is a novel antiplatelet agent which is chemically related to ticlopidine. It acts against ischaemic stroke, myocardial infraction as well as vascular disease.[1] It is reported to have exhibited clinical efficacy greater than that of aspirin, during large phase III trials.[2]
Anwendung
Hinweis zur Analyse
Sonstige Hinweise
Fußnote
Ähnliches Produkt
Signalwort
Danger
H-Sätze
Gefahreneinstufungen
Aquatic Chronic 2 - Eye Dam. 1 - Skin Corr. 1B
Lagerklassenschlüssel
8A - Combustible corrosive hazardous materials
WGK
WGK 3
Flammpunkt (°F)
Not applicable
Flammpunkt (°C)
Not applicable
Hier finden Sie alle aktuellen Versionen:
Analysenzertifikate (COA)
It looks like we've run into a problem, but you can still download Certificates of Analysis from our Dokumente section.
Wenn Sie Hilfe benötigen, wenden Sie sich bitte an Kundensupport
Besitzen Sie dieses Produkt bereits?
In der Dokumentenbibliothek finden Sie die Dokumentation zu den Produkten, die Sie kürzlich erworben haben.
Kunden haben sich ebenfalls angesehen
-
How is shipping temperature determined? And how is it related to the product storage temperature?
1 answer-
Helpful?
-
-
How can I determine the shelf life / expiration / retest date of this product?
1 answer-
If this product has an expiration or retest date, it will be shown on the Certificate of Analysis (COA, CofA). If there is no retest or expiration date listed on the product's COA, we do not have suitable stability data to determine a shelf life. For these products, the only date on the COA will be the release date; a retest, expiration, or use-by-date will not be displayed.
For all products, we recommend handling per defined conditions as printed in our product literature and website product descriptions. We recommend that products should be routinely inspected by customers to ensure they perform as expected.
For products without retest or expiration dates, our standard warranty of 1 year from the date of shipment is applicable.
For more information, please refer to the Product Dating Information document: https://www.sigmaaldrich.com/deepweb/assets/sigmaaldrich/marketing/global/documents/449/386/product-dating-information-mk.pdfHelpful?
-
Active Filters
Unser Team von Wissenschaftlern verfügt über Erfahrung in allen Forschungsbereichen einschließlich Life Science, Materialwissenschaften, chemischer Synthese, Chromatographie, Analytik und vielen mehr..
Setzen Sie sich mit dem technischen Dienst in Verbindung.