If this product has an expiration or retest date, it will be shown on the Certificate of Analysis (COA, CofA). If there is no retest or expiration date listed on the product's COA, we do not have suitable stability data to determine a shelf life. For these products, the only date on the COA will be the release date; a retest, expiration, or use-by-date will not be displayed.
For all products, we recommend handling per defined conditions as printed in our product literature and website product descriptions. We recommend that products should be routinely inspected by customers to ensure they perform as expected.
For products without retest or expiration dates, our standard warranty of 1 year from the date of shipment is applicable.
For more information, please refer to the Product Dating Information document: https://www.sigmaaldrich.com/deepweb/assets/sigmaaldrich/marketing/global/documents/449/386/product-dating-information-mk.pdf
M27301
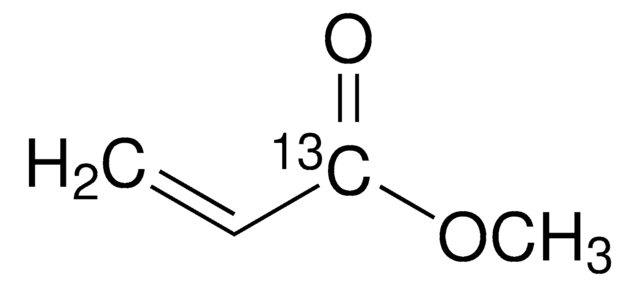
Methyl acrylate
99%, contains ≤100 ppm monomethyl ether hydroquinone as inhibitor
Sinônimo(s):
Methylacrylate
Selecione um tamanho
Selecione um tamanho
About This Item
Produtos recomendados
densidade de vapor
3 (vs air)
Nível de qualidade
pressão de vapor
67.5 mmHg ( 20 °C)
Ensaio
99%
Formulário
liquid
temperatura de autoignição
874 °F
contém
≤100 ppm monomethyl ether hydroquinone as inhibitor
Lim. expl.
14.5 %
índice de refração
n20/D 1.402 (lit.)
p.e.
80 °C (lit.)
pf
−75 °C (lit.)
densidade
0.956 g/mL at 25 °C (lit.)
temperatura de armazenamento
2-8°C
cadeia de caracteres SMILES
COC(=O)C=C
InChI
1S/C4H6O2/c1-3-4(5)6-2/h3H,1H2,2H3
chave InChI
BAPJBEWLBFYGME-UHFFFAOYSA-N
Procurando produtos similares? Visita Guia de comparação de produtos
Descrição geral
Palavra indicadora
Danger
Frases de perigo
Declarações de precaução
Classificações de perigo
Acute Tox. 3 Inhalation - Acute Tox. 4 Dermal - Acute Tox. 4 Oral - Aquatic Chronic 3 - Eye Irrit. 2 - Flam. Liq. 2 - Skin Irrit. 2 - Skin Sens. 1 - STOT SE 3
Órgãos-alvo
Respiratory system
Código de classe de armazenamento
3 - Flammable liquids
Classe de risco de água (WGK)
WGK 2
Ponto de fulgor (°F)
27.0 °F - closed cup
Ponto de fulgor (°C)
-2.8 °C - closed cup
Equipamento de proteção individual
Eyeshields, Faceshields, Gloves, type ABEK (EN14387) respirator filter
Escolha uma das versões mais recentes:
Já possui este produto?
Encontre a documentação dos produtos que você adquiriu recentemente na biblioteca de documentos.
Os clientes também visualizaram
Artigos
Our strategy is to synthesize mesoporous carbonaceous materials (“Starbons”) using mesoporous expanded starch as the precursor without the need for a templating agent.
RAFT polymerization offers living characteristics to radical polymerization, contributing versatility to reversible deactivation radical polymerization methods.
-
How can I determine the shelf life / expiration / retest date of this product?
1 answer-
Helpful?
-
-
How is shipping temperature determined? And how is it related to the product storage temperature?
1 answer-
Products may be shipped at a different temperature than the recommended long-term storage temperature. If the product quality is sensitive to short-term exposure to conditions other than the recommended long-term storage, it will be shipped on wet or dry-ice. If the product quality is NOT affected by short-term exposure to conditions other than the recommended long-term storage, it will be shipped at ambient temperature. As shipping routes are configured for minimum transit times, shipping at ambient temperature helps control shipping costs for our customers. For more information, please refer to the Storage and Transport Conditions document: https://www.sigmaaldrich.com/deepweb/assets/sigmaaldrich/marketing/global/documents/316/622/storage-transport-conditions-mk.pdf
Helpful?
-
Active Filters
Nossa equipe de cientistas tem experiência em todas as áreas de pesquisa, incluindo Life Sciences, ciência de materiais, síntese química, cromatografia, química analítica e muitas outras.
Entre em contato com a assistência técnica













![Tris[2-(dimethylamino)ethyl]amine 97%](/deepweb/assets/sigmaaldrich/product/structures/695/792/ee0ff167-22a3-43a7-83a1-6c4908adf0ae/640/ee0ff167-22a3-43a7-83a1-6c4908adf0ae.png)