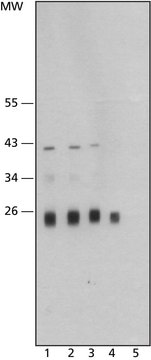
C6854
Cathepsin L from human liver
≥0.5 units/mg protein, solution
Se connecterpour consulter vos tarifs contractuels et ceux de votre entreprise/organisme
About This Item
Numéro CAS:
Numéro MDL:
Code UNSPSC :
12352200
Nomenclature NACRES :
NA.32
Produits recommandés
Source biologique
human liver
Niveau de qualité
Forme
solution
Activité spécifique
≥0.5 units/mg protein
Technique(s)
activity assay: suitable
Plage de pH
4.5—5.5
Numéro d'accès UniProt
Application(s)
pharmaceutical
Conditions d'expédition
dry ice
Température de stockage
−20°C
Informations sur le gène
human ... CTSL1(1514)
Description générale
The cathepsin L (CTSL) gene is mapped to human chromosome 9q21.33. It encodes a lysosomal cysteine proteinase that belongs to the peptidase C1 family. The enzyme is a dimer of a heavy (175 amino acids) and a light chain (42 amino acids) linked by disulfide bonds.
Application
Cathepsin L (CTSL) from human liver has been used:
- to digest mutant transforming growth factor β-induced (TGFBIp) and LC3 (autophagosomes marker) proteins in vitro
- to digest the pH-dependent fibril of a pre-melanosomal protein (Pmel17) repeat domain (RPT) isoform and study the effect of pH on sRPT aggregation kinetics by tryptophan fluorescence
- to analyze the inhibition of CTSL by small molecules or drugs in a cell-free system
Actions biochimiques/physiologiques
Cathepsin L (CTSL) exhibits endopeptidase activity and breaks peptide bonds with aromatic residues. This enzyme is functional at pH 3.0–6.5 with thiol compounds. Elastin, collagen, and α-1 protease inhibitors act as substrates for cathepsin L (CTSL). CTSL cleaves several proteins including enzymes, receptors, and transcription factors. It is involved in antigenic peptide production and controls B-cells homeostasis. It is associated with myofibril necrosis and tumor progression. Increased expression of CTSL promotes metastasis and relates to poor prognosis in cancer patients. Increased expression of CTSL is observed in breast cancer. CTSL is implicated in glioblastoma multiforme (GBM), middle east respiratory syndrome (MERS), gingival overgrowth, and severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2). It has a higher specific activity than cathepsin B and H in the degradation of a variety of physiological protein substrates.
Définition de l'unité
One unit will hydrolyze 1.0 μmole of Z-Phe-Arg-AFC per minute at pH 5.5 at 25 °C.
Forme physique
Solution in in 20 mM malonate, pH 5.5, 1 mM EDTA, and 400 mM NaCl.
Produit(s) apparenté(s)
Code de la classe de stockage
10 - Combustible liquids
Classe de danger pour l'eau (WGK)
WGK 3
Point d'éclair (°F)
Not applicable
Point d'éclair (°C)
Not applicable
Certificats d'analyse (COA)
Recherchez un Certificats d'analyse (COA) en saisissant le numéro de lot du produit. Les numéros de lot figurent sur l'étiquette du produit après les mots "Lot" ou "Batch".
Déjà en possession de ce produit ?
Retrouvez la documentation relative aux produits que vous avez récemment achetés dans la Bibliothèque de documents.
Les clients ont également consulté
Stefan Tholen et al.
Cellular and molecular life sciences : CMLS, 71(5), 899-916 (2013-07-03)
Endolysosomal cysteine cathepsins functionally cooperate. Cathepsin B (Ctsb) and L (Ctsl) double-knockout mice die 4 weeks after birth accompanied by (autophago-) lysosomal accumulations within neurons. Such accumulations are also observed in mouse embryonic fibroblasts (MEFs) deficient for Ctsb and Ctsl.
M Kathryn Liszewski et al.
Immunity, 39(6), 1143-1157 (2013-12-10)
Complement is viewed as a critical serum-operative component of innate immunity, with processing of its key component, C3, into activation fragments C3a and C3b confined to the extracellular space. We report here that C3 activation also occurred intracellularly. We found
Dexter N Dean et al.
Biochimica et biophysica acta. Proteins and proteomics, 1867(10), 961-969 (2019-02-05)
The pre-melanosomal protein (Pmel17) aggregates within melanosomes to form functional amyloid fibrils that facilitate melanin polymerization. The repeat domain (RPT) of Pmel17 fibrillates under strict acidic melanosomal pH. Alternative splicing results in a shortened repeat domain (sRPT), which also forms
Bailey Miller et al.
Journal of natural products, 77(1), 92-99 (2013-12-25)
A number of marine natural products are potent inhibitors of proteases, an important drug target class in human diseases. Hence, marine cyanobacterial extracts were assessed for inhibitory activity to human cathepsin L. Herein, we have shown that gallinamide A potently
Gustavo E Chavarria et al.
European journal of medicinal chemistry, 58, 568-572 (2012-11-22)
Kinetic analysis of the mode of inhibition of cathepsin L by KGP94, a lead compound from a privileged library of functionalized benzophenone thiosemicarbazone derivatives, demonstrated that it is a time-dependent, reversible, and competitive inhibitor of the enzyme. These results are
Notre équipe de scientifiques dispose d'une expérience dans tous les secteurs de la recherche, notamment en sciences de la vie, science des matériaux, synthèse chimique, chromatographie, analyse et dans de nombreux autres domaines..
Contacter notre Service technique