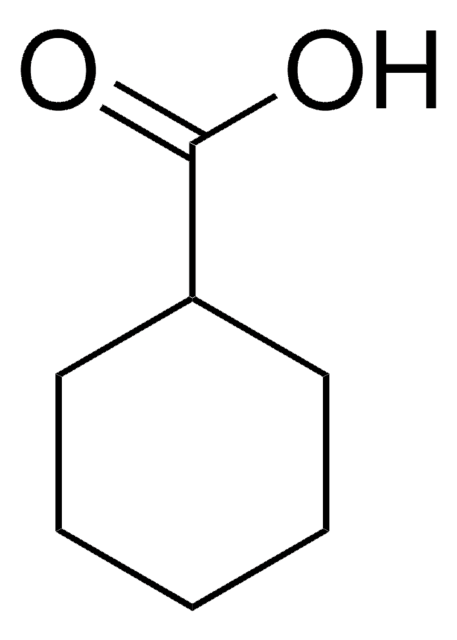
P37602
5-Phenylvaleric acid
99%
Synonym(s):
5-Phenylpentanoic acid
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Linear Formula:
C6H5CH2CH2CH2CH2COOH
CAS Number:
Molecular Weight:
178.23
Beilstein:
2049062
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Quality Level
Assay
99%
form
crystals
bp
177-178 °C/13 mmHg (lit.)
mp
58-60 °C (lit.)
SMILES string
OC(=O)CCCCc1ccccc1
InChI
1S/C11H14O2/c12-11(13)9-5-4-8-10-6-2-1-3-7-10/h1-3,6-7H,4-5,8-9H2,(H,12,13)
InChI key
BYHDDXPKOZIZRV-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
D M Chung et al.
International journal of biological macromolecules, 29(4-5), 243-250 (2001-11-24)
From a set of mixed carbon sources, 5-phenylvaleric acid (PV) and octanoic acid (OA), polyhydroxyalkanoic acid (PHA) was separately accumulated in the two pseudomonads Pseudomonas putida BM01 and Pseudomonas citronellolis (ATCC 13674) to investigate any structural difference between the two
Christopher Jewell et al.
Drug metabolism and disposition: the biological fate of chemicals, 35(11), 2015-2022 (2007-08-01)
The capacity of human, minipig, and rat skin and liver subcellular fractions to hydrolyze the anesthetic ester procaine was compared with carboxylesterase substrates 4-methylumbelliferyl-acetate, phenylvalerate, and para-nitrophenylacetate and the arylesterase substrate phenylacetate. Rates of procaine hydrolysis by minipig and human
Marie-Paule Gonthier et al.
Free radical biology & medicine, 35(8), 837-844 (2003-10-15)
Procyanidins are major dietary polyphenols made of elementary flavan-3-ol (epi)catechin units. They have antioxidant properties and may contribute to health benefits in humans, but little is known about their metabolic fate. We compared here the metabolism of procyanidin dimer B3
I Calis et al.
Journal of natural products, 62(8), 1101-1105 (1999-09-10)
(3R)-O-beta-D-Glucopyranosyloxy-5-phenylvaleric acid (1), (3R)-O-beta-D-glucopyranosyloxy-5-phenylvaleric acid n-butyl ester (2), and a new dihydrochalcone diglycoside 4'-O-[beta-D-glucopyranosyl-(1-->6)-glucopyranosyl]oxy-2'-hydroxy-3', 6'-dimethoxydihydrochalcone (3), together with six known flavonoid glycosides [kaempferol-3-O-beta-D-glucopyranoside (= astragalin) (4), kaempferol-3-O-beta-D-galactopyranoside (5), quercetin-3-O-beta-D-glucopyranoside (= isoquercitrin) (6), quercetin-3-O-beta-D-galactopyranoside (= hyperoside) (7), quercetin-3-O-(2''-O-galloyl)-beta-D-glucopyranoside (8), and quercetin-3-O-beta-D-glucuronopyranoside
I-Ching Ho et al.
International journal of biological macromolecules, 40(2), 112-118 (2006-08-22)
PHAs (poly-3-hydroxyalkanoates) obtained by Pseudomonas oleovorans grown with mixed carbon sources were investigated. Mixed carbon sources were sodium octanoate/undecylenic acid and sodium octanoate/5-phenylvaleric acid. Effect of carbon source in pre-culture on PHAs structure was investigated. Main fermentation was conducted with
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service