Select a Size
| Pack Size | SKU | Availability | Price |
|---|---|---|---|
| 50 mg | Available to ship TODAYfromMILWAUKEE | $344.00 |
About This Item
form
powder
Quality Segment
color
white
solubility
H2O: 10 mg/mL
storage temp.
2-8°C
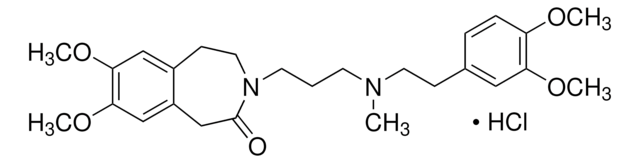
SMILES string
Cl[H].Cl[H].COc1cc(cc(OC)c1OC)C(=O)OCCCN2CCCN(CCCOC(=O)c3cc(OC)c(OC)c(OC)c3)CC2
InChI
1S/C31H44N2O10.2ClH/c1-36-24-18-22(19-25(37-2)28(24)40-5)30(34)42-16-8-12-32-10-7-11-33(15-14-32)13-9-17-43-31(35)23-20-26(38-3)29(41-6)27(21-23)39-4;;/h18-21H,7-17H2,1-6H3;2*1H
InChI key
VILIWRRWAWKXRW-UHFFFAOYSA-N
Gene Information
human ... ADORA1(134), ADORA2A(135), ADORA2B(136), ADORA3(140)
General description
Application
Biochem/physiol Actions
1 of 1
This Item | |||
|---|---|---|---|
| form powder | form powder | form powder | form powder |
| Quality Level 200 | Quality Level 100 | Quality Level 100 | Quality Level 200 |
| storage temp. 2-8°C | storage temp. 2-8°C | storage temp. 2-8°C | storage temp. - |
| solubility H2O: 10 mg/mL | solubility H2O: >10 mg/mL | solubility H2O: 20 mg/mL, clear | solubility H2O: 50 mg/mL |
| color white | color white | color white to beige | color white |
| Gene Information human ... ADORA1(134), ADORA2A(135), ADORA2B(136), ADORA3(140) | Gene Information - | Gene Information human ... SCN4A(6329) | Gene Information human ... HTR1A(3350), HTR1B(3351), HTR1D(3352), HTR1E(3354), HTR1F(3355), HTR2A(3356), HTR2B(3357), HTR2C(3358), HTR3A(3359), HTR3B(9177), HTR3C(170572), HTR3D(200909), HTR3E(285242), HTR4(3360), HTR5A(3361), HTR5B(645694), HTR6(3362), HTR7(3363), SLC6A2(6530), SLC6A4(6532) |
signalword
Warning
hcodes
Hazard Classifications
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
target_organs
Respiratory system
Storage Class
11 - Combustible Solids
wgk
WGK 2
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Global Trade Item Number
| SKU | GTIN |
|---|---|
| D5294-50MG | 04061832081922 |
| D5294-100MG | 04061832081915 |