429066
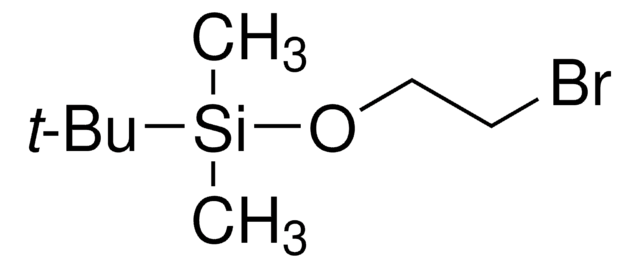
(3-溴丙氧基)-叔丁基二甲基硅烷
97%
同義詞:
(3-Bromopropoxy)(1,1-dimethylethyl)dimethylsilane, 1-((tert-Butyldimethylsilyl)oxy)-3-bromopropane, 1-Bromo-3-(tert-butyldimethylsiloxy)propane, 1-Bromo-3-[(tert-butyldimethylsilanyl)oxy]propane, 1-Bromo-3-[(tert-butyldimethylsilyl)oxy]propane, 3-((tert-Butyldimethylsilyl)oxy)-1-bromopropane
About This Item
推薦產品
化驗
97%
形狀
liquid
包含
sodium carbonate as stabilizer
折射率
n20/D 1.451 (lit.)
bp
182 °C (lit.)
密度
1.093 g/mL at 25 °C (lit.)
官能基
bromo
儲存溫度
2-8°C
SMILES 字串
CC(C)(C)[Si](C)(C)OCCCBr
InChI
1S/C9H21BrOSi/c1-9(2,3)12(4,5)11-8-6-7-10/h6-8H2,1-5H3
InChI 密鑰
QGMROEZDWJTIDW-UHFFFAOYSA-N
訊號詞
Warning
危險聲明
危險分類
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
標靶器官
Respiratory system
儲存類別代碼
10 - Combustible liquids
水污染物質分類(WGK)
WGK 3
閃點(°F)
185.0 °F - closed cup
閃點(°C)
85 °C - closed cup
客戶也查看了
Active Filters
我們的科學家團隊在所有研究領域都有豐富的經驗,包括生命科學、材料科學、化學合成、色譜、分析等.
聯絡技術服務