Wybierz wielkość
1370,00 zł
1370,00 zł
Informacje o tej pozycji
Przejdź do
form
lyophilized powder
specific activity
25-75 units/mg protein
mol wt
~128 kDa (by gel filtration)
composition
Protein, ≥60% biuret
storage temp.
−20°C
Quality Level
1 of 4
Ta pozycja | G4509 | G0774 | G9637 |
|---|---|---|---|
| specific activity 25-75 units/mg protein | specific activity 40-100 units/mg protein | specific activity ≥75 units/mg protein (biuret) | specific activity 40-80 units/mg solid, pH 8.1 |
| form lyophilized powder | form lyophilized powder | form buffered aqueous solution | form lyophilized powder |
| mol wt ~128 kDa (by gel filtration) | mol wt - | mol wt - | mol wt ~76 kDa (gel filtration) |
| storage temp. −20°C | storage temp. −20°C | storage temp. 2-8°C | storage temp. 2-8°C |
| Quality Level 200 | Quality Level 200 | Quality Level 200 | Quality Level 200 |
| composition Protein, ≥60% biuret | composition Protein, ≥50% biuret | composition - | composition - |
General description
Glycerol kinase (GK) is part of the FGGY carbohydrate kinase family.[1]
Michaelis constants : 4.4 x 10-5M (Glycerol), 4.3 x 10-4M (ATP)
Inhibitors : p-Chloromercuribenzoate, heavy metal ions (Pb++, Fe++, Hg++, Ag+)
Optimum pH : 9.8 (G-3-PDH system), 7.8 (G-3-P oxidase system) Optimum temperature : 500C
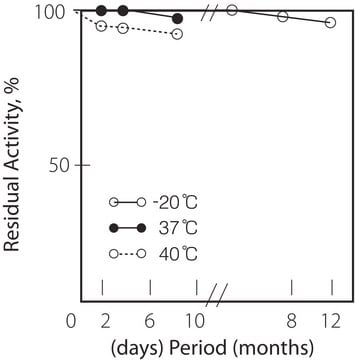
pH Stability : pH 5.5 x 10.0 (25oC, 20hr)
Thermal stability : below 40oC (pH 7.5, 15min)
Substrate specificity : This enzyme catalyzes the stereospecific transfer of the terminal
phosphoryl moiety of ATP to one of the primary hydroxyl group of
glycerol, forming sn-glycerol-3-P. The enzyme has the highest
specificity for glycerol, and also phosphorylates dihydroxyacetone
and glyceraldehyde (Table 1,2). Mg++ is essentially required for the
reaction.
Application
- for determining the kinetic characteristics of human and trypanosomatid phosphofructokinases using an enzyme-linked kinetic assay.[2]
- to study the effect of sugar in fluorescence emission.[3]
- in 2-Arachidonoylglycerol-based fluorescence assay for DH-463, a fluorescent activity-based probe for monoacylglycerol lipase.[4]
Biochem/physiol Actions
Physical form
Other Notes
signalword
Danger
hcodes
pcodes
Hazard Classifications
Resp. Sens. 1
Klasa składowania
11 - Combustible Solids
wgk
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
Eyeshields, Gloves, type N95 (US)
Wybierz jedną z najnowszych wersji:
Masz już ten produkt?
Dokumenty związane z niedawno zakupionymi produktami zostały zamieszczone w Bibliotece dokumentów.
Powiązane treści
Active Filters
Nasz zespół naukowców ma doświadczenie we wszystkich obszarach badań, w tym w naukach przyrodniczych, materiałoznawstwie, syntezie chemicznej, chromatografii, analityce i wielu innych dziedzinach.
Skontaktuj się z zespołem ds. pomocy technicznej